Sublimation Examples

Sublimation Process Explained Learn what sublimation is and see 22 examples of sublimation in daily life, such as dry ice, camphor, iodine, and snow. sublimation is when a solid changes directly into a gas without melting into a liquid. Dry ice is the solid form of.
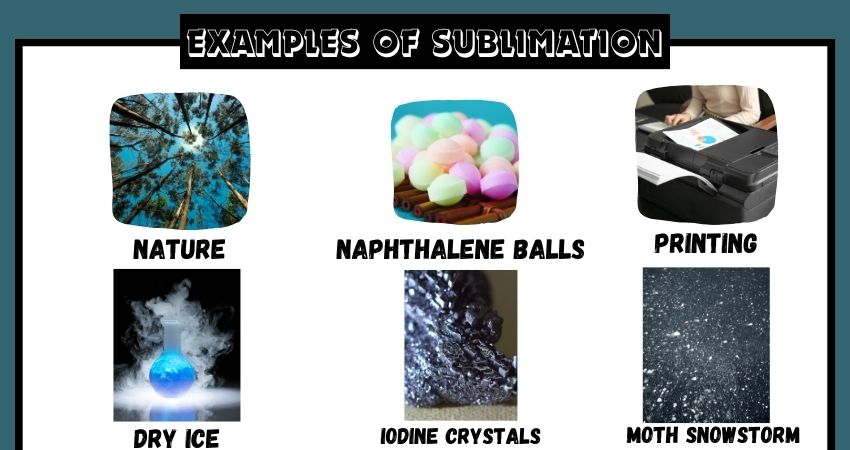
25 Examples Of Sublimation Vivid Examples Learn what sublimation is, how to sublimate a substance, and what are the examples and applications of sublimation. sublimation is the transition of a substance from solid to gas without liquid phase, and it is used in freeze drying, printing, air fresheners, and more. Learn what sublimation is and how it occurs in nature and everyday objects. see examples of sublimation in dry ice, water cycle, mothballs, dye sublimation printing, forensics, perfume tablets and space. Notable examples include sublimation of dry ice at room temperature and atmospheric pressure, and that of solid iodine with heating. the reverse process of sublimation is deposition (also called desublimation), in which a substance passes directly from a gas to a solid phase, without passing through the intermediate liquid state. Learn about sublimation, a physical process where a substance transitions directly from solid to gas without liquid. discover 25 examples of sublimation in various substances and everyday scenarios, such as dry ice, naphthalene, camphor, iodine, snow, and more.
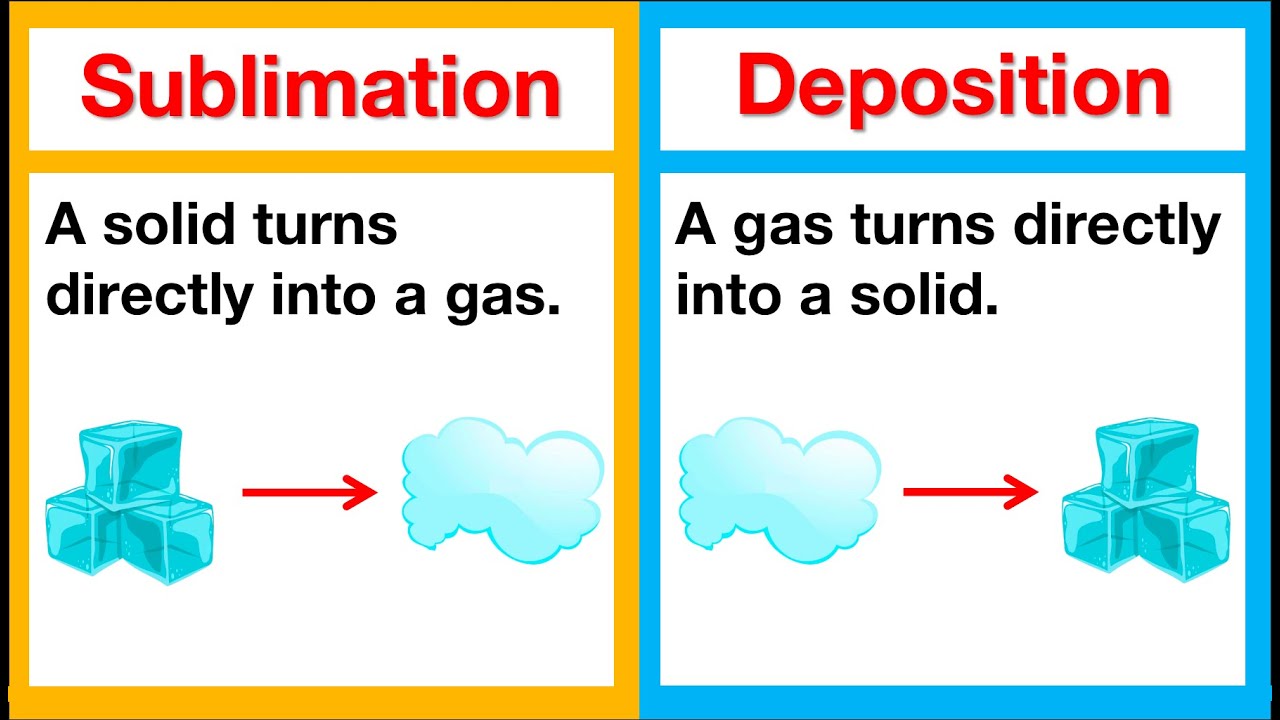
Sublimation Examples Notable examples include sublimation of dry ice at room temperature and atmospheric pressure, and that of solid iodine with heating. the reverse process of sublimation is deposition (also called desublimation), in which a substance passes directly from a gas to a solid phase, without passing through the intermediate liquid state. Learn about sublimation, a physical process where a substance transitions directly from solid to gas without liquid. discover 25 examples of sublimation in various substances and everyday scenarios, such as dry ice, naphthalene, camphor, iodine, snow, and more. Sublimation is the conversion of a substance from solid to gas without becoming liquid. learn about the phenomenon, its causes, and examples such as dry ice and freeze drying of food. This detailed and simple guide will help you learn about sublimation, its process, examples, applications, and importance in science and industry. Here are some well known substances that act as sublimands: iodine (i₂) → on heating, iodine crystals turn directly into purple vapor. dry ice (solid co₂) → sublimes at room temperature without melting, producing dense white vapor. naphthalene → found in mothballs; sublimates slowly at room temperature. The video below shows the sublimation and deposition of iodine. the beaker containing iodine is covered with a watch glass that contains ice. when the iodine is heated it sublimes. when iodine vapors cool, deposition occurs. deposition is the change of state from a gas to a solid.
Comments are closed.