Structure Of Diamond %d1%80%d1%9f %d1%9c Arrangement Of Carbon Atoms In Diamond

Describe The Structure Of Diamond Draw A Simple Diagram To Show The Each carbon atom joins four other carbon atoms in regular tetrahedrons (triangular prisms). based on the cubic form and its highly symmetrical arrangement of atoms, diamond crystals can develop into several different shapes, known as 'crystal habits'. The most notable material with a diamond cubic structure is, of course, carbon (in diamonds). the diamond cubic structure is tetrahedrally coordinated, which means that each atom has 4 bonds.

Structure Of Diamond And Uses Pdf Silicon Dioxide Graphite Diamond's cubic structure is in the fd 3 m space group (space group 227), which follows the face centered cubic bravais lattice. Diamonds are carbon allotropes that have a three dimensional lattice structure. each carbon atom makes strong covalent bonds with four nearby carbon atoms, forming a tetrahedral configuration. diamonds have extraordinary hardness and stability due to this configuration. The carbon atoms in diamond are said to generate strong chemical bonds with the other four carbon atoms, resulting in a perfect tetrahedron structure that extends throughout the crystal. The structure is not a bravais lattice by itself because there are two types of lattice points with different environments. but when we choose a proper perspective, we can see that the underlying structure is actually a fcc structure with a two atomic basis.

Carbon Atoms Diamond Hi Res Stock Photography And Images Alamy The carbon atoms in diamond are said to generate strong chemical bonds with the other four carbon atoms, resulting in a perfect tetrahedron structure that extends throughout the crystal. The structure is not a bravais lattice by itself because there are two types of lattice points with different environments. but when we choose a proper perspective, we can see that the underlying structure is actually a fcc structure with a two atomic basis. The crystalline structure of the diamond is a face centered cubic crystal structure or fcc. in this crystalline structure, each carbon atom is joined to four more carbon atoms in a regular tetrahedral pattern. Crystal diamond is defined as a structure composed of sp³ hybridized carbon atoms arranged in a three dimensional network of purely covalent bonds, typically exhibiting a cubic system due to the periodic repetition of c–c bonds. The rigid network of carbon atoms, held together by strong covalent bonds, makes diamond very hard. this makes it useful for cutting tools, such as diamond tipped glass cutters and oil rig. The crystal structure of diamond is equivalent to a face centred cubic (fcc) lattice, with a basis of two identical carbon atoms: one at (0, 0, 0) and the other at (1 4, 1 4, 1 4), where the coordinates are given as fractions along the cube sides.
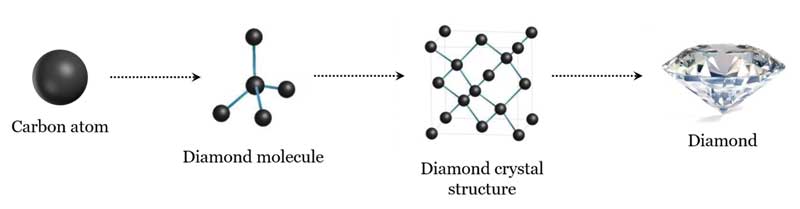
Carbon Structure Of Diamond The crystalline structure of the diamond is a face centered cubic crystal structure or fcc. in this crystalline structure, each carbon atom is joined to four more carbon atoms in a regular tetrahedral pattern. Crystal diamond is defined as a structure composed of sp³ hybridized carbon atoms arranged in a three dimensional network of purely covalent bonds, typically exhibiting a cubic system due to the periodic repetition of c–c bonds. The rigid network of carbon atoms, held together by strong covalent bonds, makes diamond very hard. this makes it useful for cutting tools, such as diamond tipped glass cutters and oil rig. The crystal structure of diamond is equivalent to a face centred cubic (fcc) lattice, with a basis of two identical carbon atoms: one at (0, 0, 0) and the other at (1 4, 1 4, 1 4), where the coordinates are given as fractions along the cube sides.
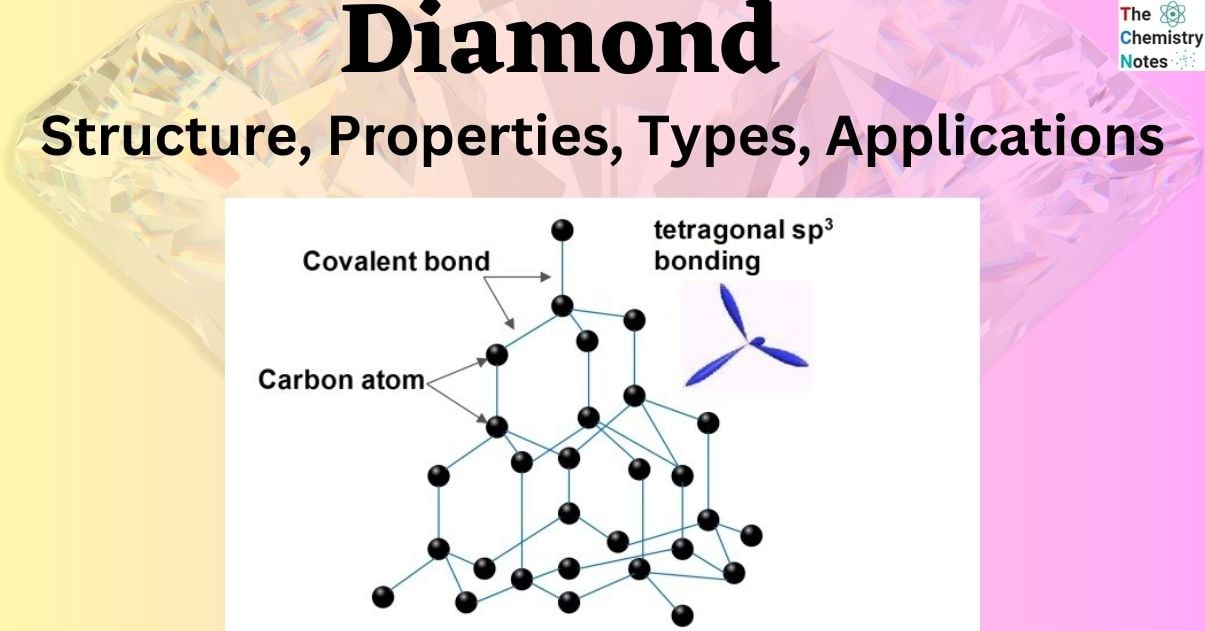
Carbon Structure Of Diamond The rigid network of carbon atoms, held together by strong covalent bonds, makes diamond very hard. this makes it useful for cutting tools, such as diamond tipped glass cutters and oil rig. The crystal structure of diamond is equivalent to a face centred cubic (fcc) lattice, with a basis of two identical carbon atoms: one at (0, 0, 0) and the other at (1 4, 1 4, 1 4), where the coordinates are given as fractions along the cube sides.
Comments are closed.