Carbon Structure Of Diamond

Carbon Diamond Structure Briefly Distinguish Between The Graphite And In diamond, each carbon shares electrons with four other carbon atoms forming four single bonds. in the diagram some carbon atoms only seem to be forming two bonds (or even one bond), but that's not really the case. we are only showing a small bit of the whole structure. A diamond’s crystal structure is a face centered cubic or fcc lattice. in typical tetrahedrons (triangular prisms), each carbon atom connects four other carbon atoms.
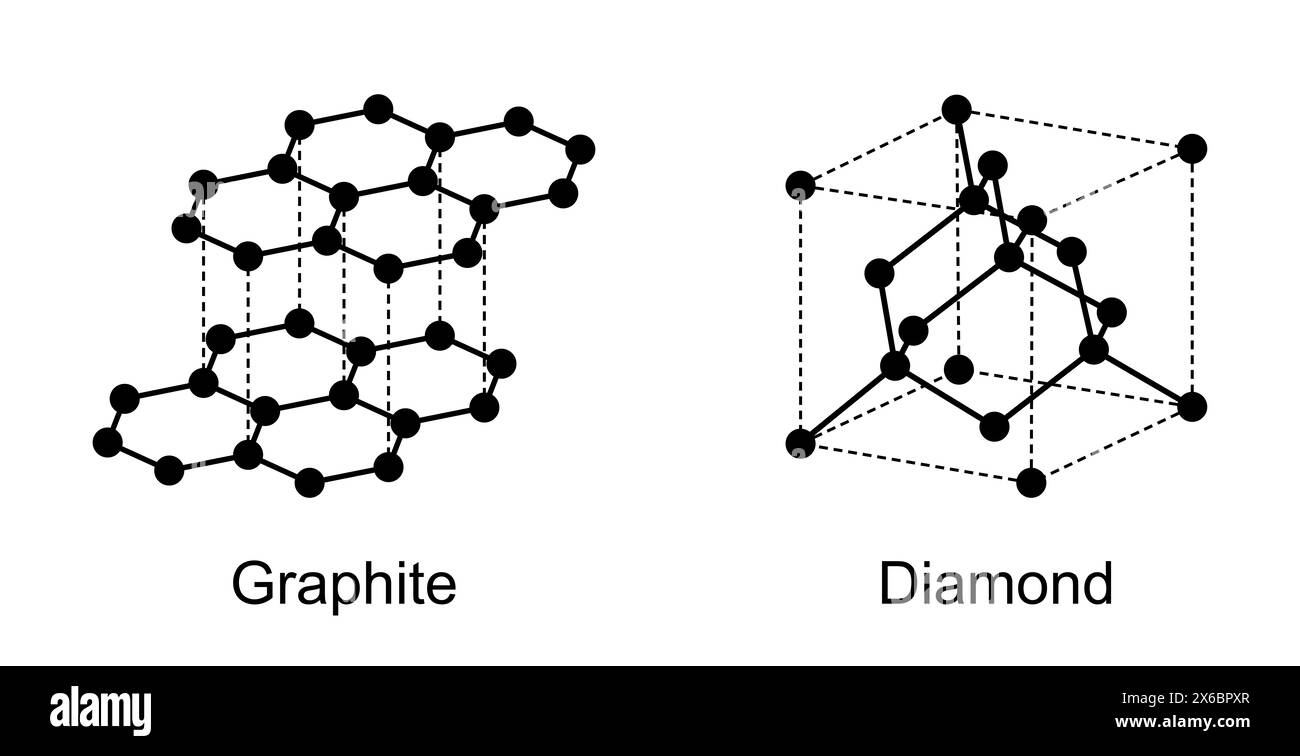
Carbon Diamond Structure Briefly Distinguish Between The Graphite And The crystal structure of a diamond is a face centered cubic or fcc lattice. each carbon atom joins four other carbon atoms in regular tetrahedrons (triangular prisms). Diamond is a mineral form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. diamond is a tasteless, odorless, strong, brittle solid, a poor conductor of electricity, colorless in pure form, and insoluble in water. Diamond is a type of carbon that has its atoms arranged in a diamond cubic crystal structure. another solid form of carbon known as graphite is the chemically stable form of carbon at ambient temperature and pressure, but diamond almost never transforms to it. Diamond is one of the hardest forms of carbon and is mainly used for making beautiful gemstones. the structure of the diamond is tetrahedral with each of the carbon atoms joined to the other four carbon atoms in a regular tetrahedral pattern.
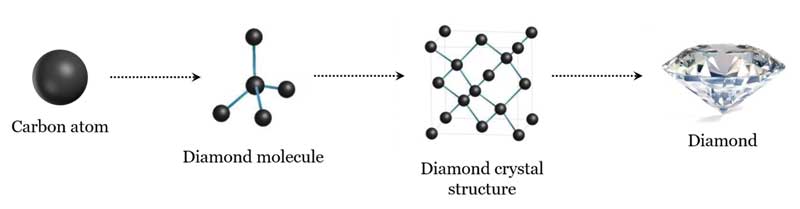
Carbon Structure Of Diamond Diamond is a type of carbon that has its atoms arranged in a diamond cubic crystal structure. another solid form of carbon known as graphite is the chemically stable form of carbon at ambient temperature and pressure, but diamond almost never transforms to it. Diamond is one of the hardest forms of carbon and is mainly used for making beautiful gemstones. the structure of the diamond is tetrahedral with each of the carbon atoms joined to the other four carbon atoms in a regular tetrahedral pattern. Each carbon atom in diamond forms four equally spaced bonds with its neighbors, creating a shape called a tetrahedron, essentially a pyramid with a triangular base. these are single covalent bonds, meaning each pair of atoms shares electrons directly. We now know that diamond is an allotrope of the element carbon arranged in a cubic lattice with sp 3 hybrid bonds. Six atoms fall on the middle of each of the six cube faces, showing two bonds. out of eight cube corners, four atoms bond to an atom within the cube. the other four bond to adjacent cubes of the crystal. Use our revision notes to learn about diamond structure for your chemistry gcse exam. explain its properties including melting boiling point and conductivity.
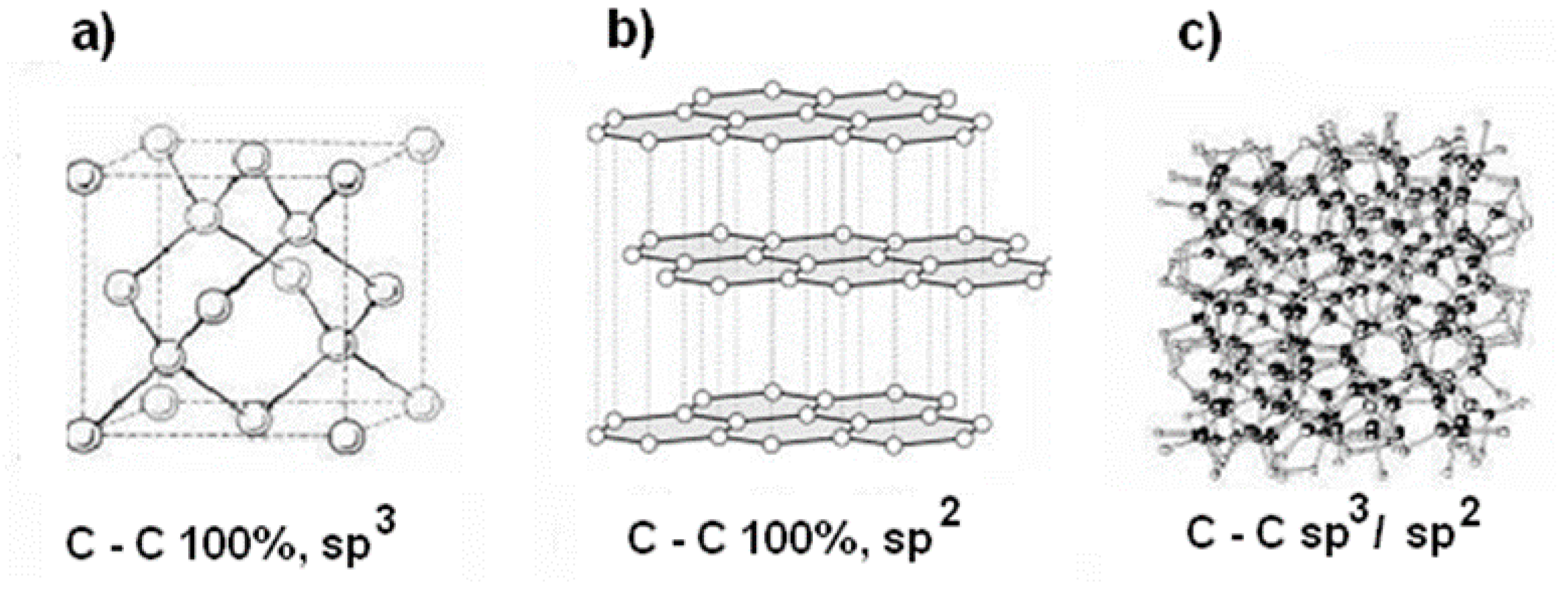
Carbon Structure Of Diamond Each carbon atom in diamond forms four equally spaced bonds with its neighbors, creating a shape called a tetrahedron, essentially a pyramid with a triangular base. these are single covalent bonds, meaning each pair of atoms shares electrons directly. We now know that diamond is an allotrope of the element carbon arranged in a cubic lattice with sp 3 hybrid bonds. Six atoms fall on the middle of each of the six cube faces, showing two bonds. out of eight cube corners, four atoms bond to an atom within the cube. the other four bond to adjacent cubes of the crystal. Use our revision notes to learn about diamond structure for your chemistry gcse exam. explain its properties including melting boiling point and conductivity.
Comments are closed.