Structure Ipec Federation
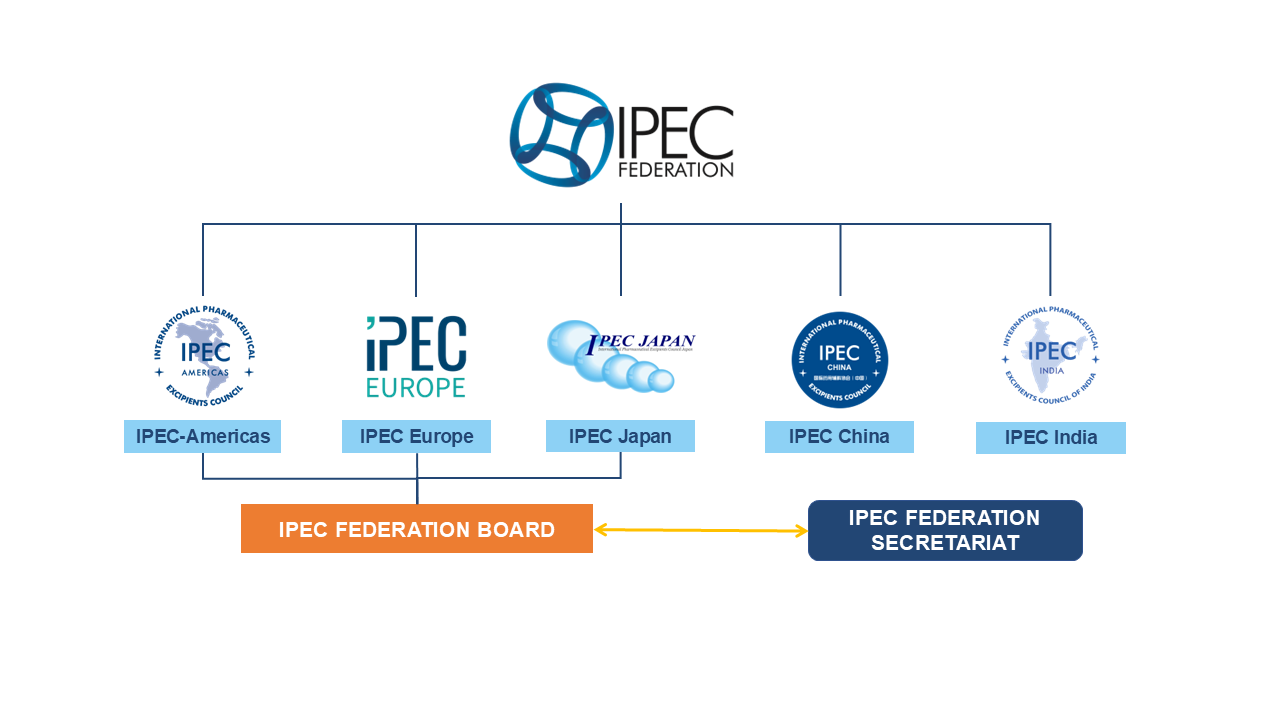
Structure Ipec Federation Ipec federation international pharmaceutical excipients council – ipec federation. Key objectives for the federation are harmonization of standards, promoting supply chain security, and the further development of third party certification, as well as providing a source of advice and expertise for other stakeholders on excipients.

Structure Ipec Federation The meeting was led by mr kevin hughes, president, ipec federation. other officers include, priscilla zawislak, vice president, janeen skutnik wilkinson, representing ipec americas, and hiroshi watanabe, treasurer, representing ipec japan as well as others from around the globe. The tripec structure evolves into the ipec federation, a global association consisting of regional associations organised to promote quality in excipients and legally registered in belgium. Profile of ipec federation from the yearbook of international organizations, a service of the uia. Ipec europe develops and publishes guidelines to promote the best use of excipients in medicines as a means of improving patient safety. since 1992, ipec europe brings together producers, distributors and users of pharmaceutical excipients to exchange good practices and develop harmonised standards.

Ipec Federation Profile of ipec federation from the yearbook of international organizations, a service of the uia. Ipec europe develops and publishes guidelines to promote the best use of excipients in medicines as a means of improving patient safety. since 1992, ipec europe brings together producers, distributors and users of pharmaceutical excipients to exchange good practices and develop harmonised standards. Formally created in 2010 in cannes (france), the federation provides a unified voice to promote the best use of excipients in medicines as a means of improving patient treatment and safety. Ipec federation is a global organisation that promotes quality and safety in pharmaceutical excipients. excipients play a critical role in the manufacture of medicines by helping to preserve the efficacy, safety, and stability of active pharmaceutical ingredients (apis), and helping to ensure that they deliver their promised benefits to patients. Welcome to our first 2024 issue of the ipec federation bulletin, which we hope you will find interesting. in this edition, we delve into the priority objectives for 2024 and regional updates. Ipec qualification of excipients guide – version 2 – 2020 ipec gmp certification scheme and cb qualification guide – version 1 – 2020 ipec excipient composition guide – version 2 – 2020 pda ipec technical report 54.6 formalized risk assessment for excipients – version 1 – 2019.
Comments are closed.