Sterilization Wfhss Guidelines

Wfhss Guidelines Saturated steam sterilization (also referred to as moist heat sterilization or steam sterilization), is the preferred sterilization method for non heat sensitive devices. The 26th edition of the world sterilization congress took place on 3rd to 6th december 2025 in hong kong. the theme of the 2025 world sterilization congress was “bring the sterilization science to the next level”.
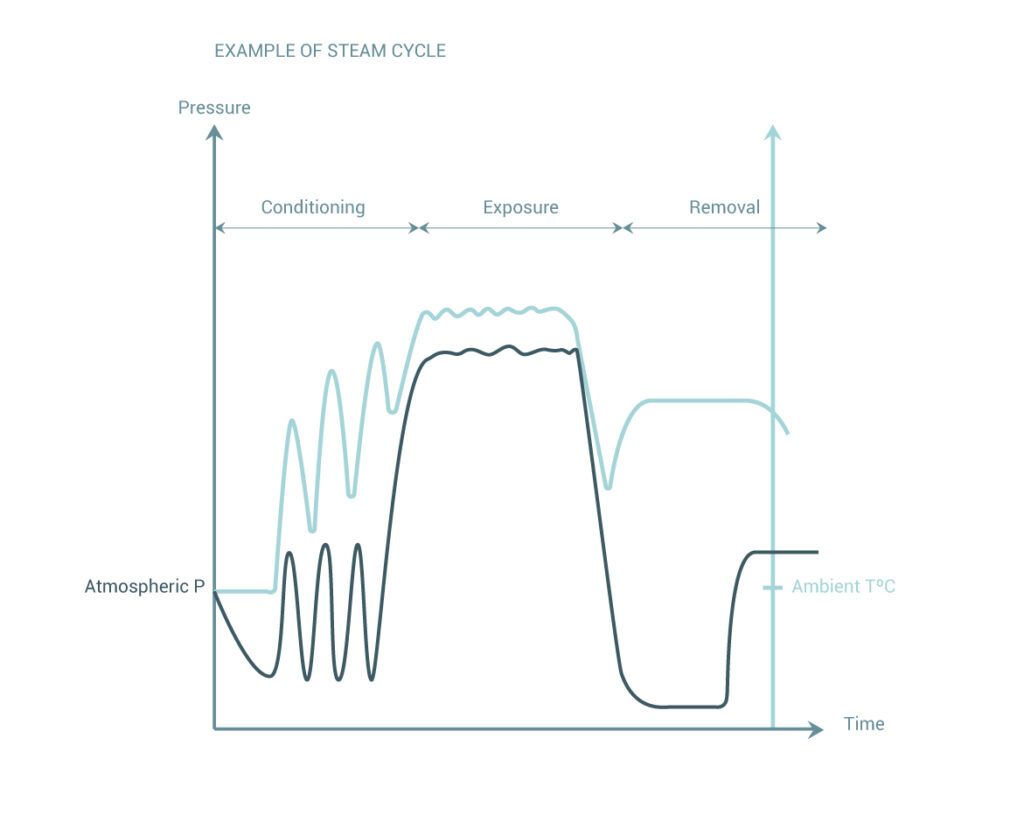
Sterilization Wfhss Guidelines Official website of the world forum for hospital sterile supply (wfhss) hosting and linking to national and regional associations for hospital sterile supply managers and professionals. This document discusses validation of decontamination processes for medical devices, including steam sterilization, sterilization with formaldehyde, sterilization with ethylene oxide, and cleaning and disinfection processes. They are intended to provide guidance and the state of the art recommendations from an academic world society focused on science but their purpose is not to supersede local regulation, standards or guidelines. Download recommendations by the quality task group (99) – recommendations for validation preparation of steam sterilization processes in large sterilizers – part 2 – 2016 – central service.
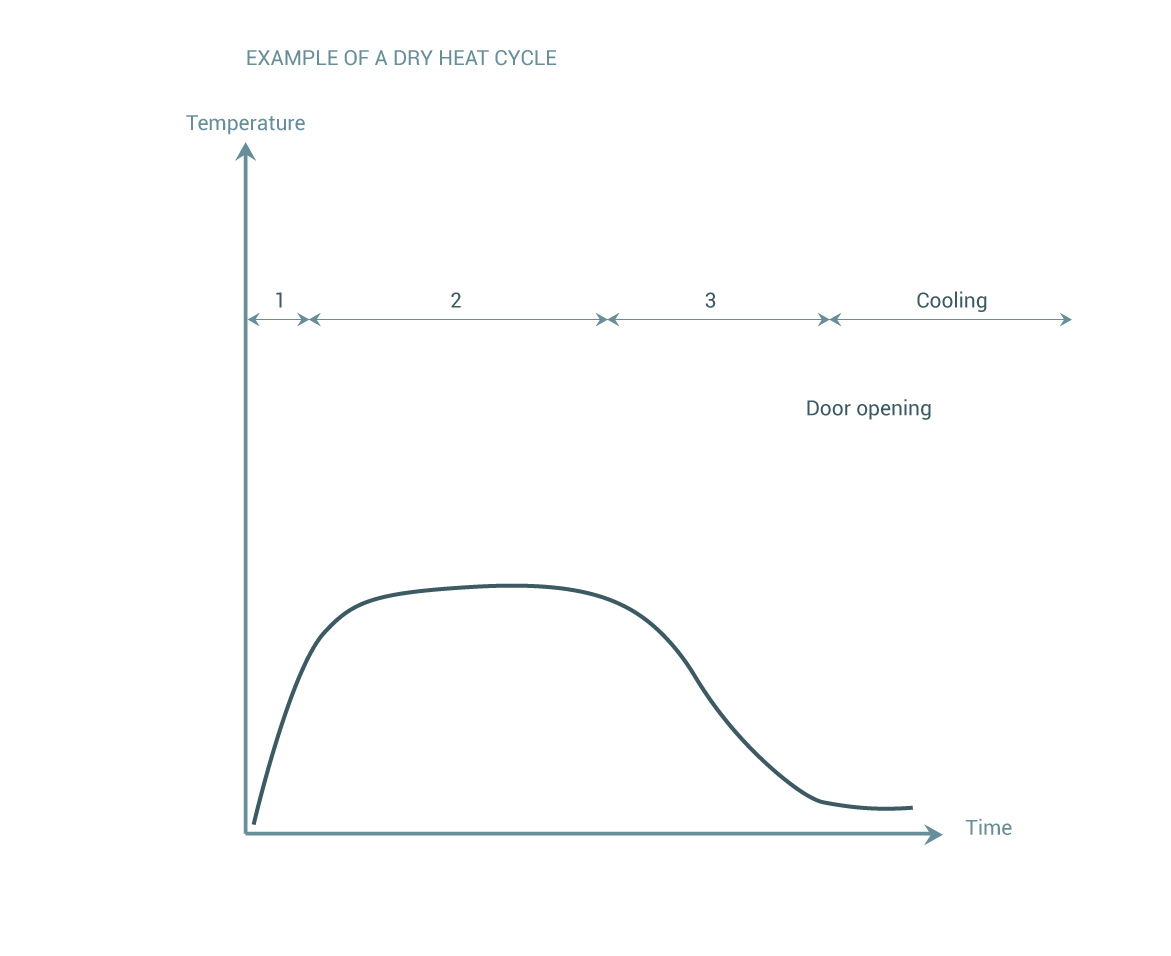
Sterilization Wfhss Guidelines They are intended to provide guidance and the state of the art recommendations from an academic world society focused on science but their purpose is not to supersede local regulation, standards or guidelines. Download recommendations by the quality task group (99) – recommendations for validation preparation of steam sterilization processes in large sterilizers – part 2 – 2016 – central service. Diseases such as hepatitis b, known to be transmitted through contaminated surgical instruments, stimulated the need for stricter guidelines for disinfection and sterilization. Diseases such as hepatitis b, known to be transmitted through contaminated surgical instruments, stimulated the need for stricter guidelines for disinfection and sterilization. Guidance may encompass all reprocessing steps or sterilization methods within a single document (e.g., wfhss) or be segmented across multiple documents, as with aami’s recommended practices. Decontamination and reprocessing of medical devices for health care facilities (who guideline persian version) regulation (eu) 2017 745 of the european parliament and of the council of 5 april 2017 on medical devices.

Sterilization Wfhss Guidelines Diseases such as hepatitis b, known to be transmitted through contaminated surgical instruments, stimulated the need for stricter guidelines for disinfection and sterilization. Diseases such as hepatitis b, known to be transmitted through contaminated surgical instruments, stimulated the need for stricter guidelines for disinfection and sterilization. Guidance may encompass all reprocessing steps or sterilization methods within a single document (e.g., wfhss) or be segmented across multiple documents, as with aami’s recommended practices. Decontamination and reprocessing of medical devices for health care facilities (who guideline persian version) regulation (eu) 2017 745 of the european parliament and of the council of 5 april 2017 on medical devices.

Sterilization Wfhss Guidelines Guidance may encompass all reprocessing steps or sterilization methods within a single document (e.g., wfhss) or be segmented across multiple documents, as with aami’s recommended practices. Decontamination and reprocessing of medical devices for health care facilities (who guideline persian version) regulation (eu) 2017 745 of the european parliament and of the council of 5 april 2017 on medical devices.
Comments are closed.