Sterile Storage

Sterile Storage Cabinets Storage Cabinet Nanotechnology Products Npd Learn the essential aorn, aami, ashrae sterile storage standards for optimal environmental control. ensure compliance and patient safety. Learn sterile instrument storage standards from joint commission, cdc, aami & osha. temperature, humidity, shelving & handling requirements for compliance.

Sterile Storage Cabinet The central processing area (s) ideally should be divided into at least three areas: decontamination, packaging, and sterilization and storage. physical barriers should separate the decontamination area from the other sections to contain contamination on used items. Sterile storage requirements ntegrity of the packages and re duce the potential for contamination. the sterile storage area, preferably an enclosed room, should be easily ac cessible rom the sterilization cooling, breakout, and case cart staging areas. it should be designated as a semi r. Most ebgs agree that sterile supply areas must be clean, well ventilated and protect supplies from contamination, moisture, dust, temperature extremes, and humidity extremes. These systems serve as a centralized storage location for sterile supplies, surgical instrument kits, and clean items that need controlled handling and traceability across departments.
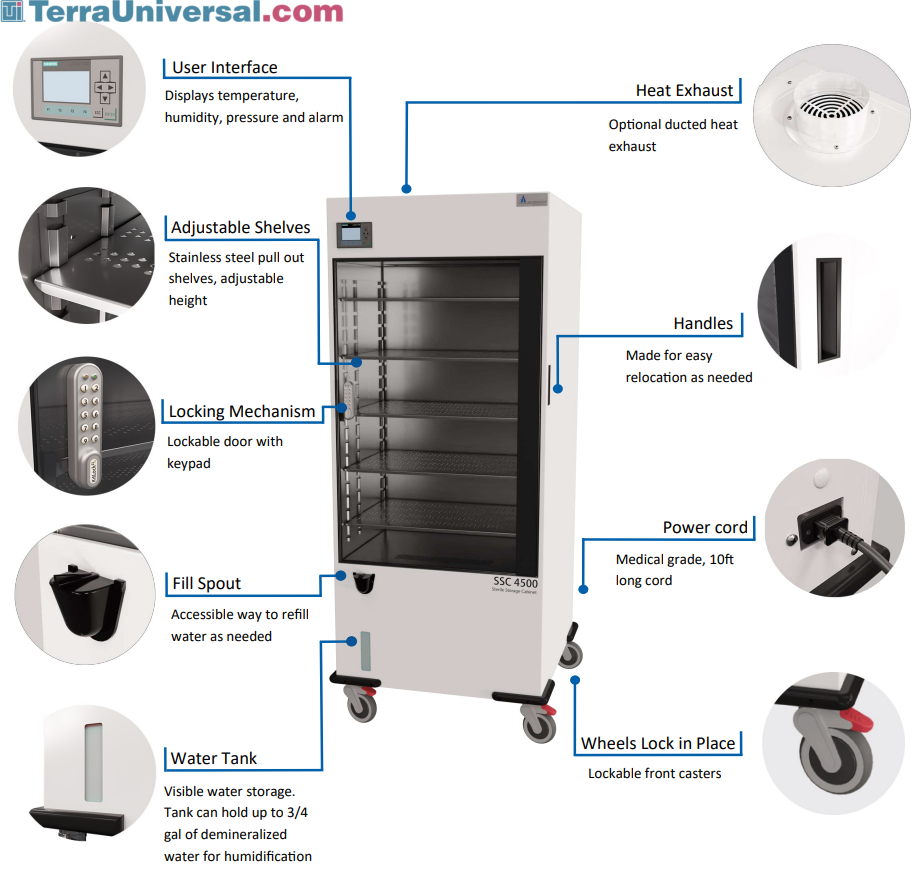
Sterile Storage Cabinet Most ebgs agree that sterile supply areas must be clean, well ventilated and protect supplies from contamination, moisture, dust, temperature extremes, and humidity extremes. These systems serve as a centralized storage location for sterile supplies, surgical instrument kits, and clean items that need controlled handling and traceability across departments. Where can i find information on sterilization for dental practices? please see ansi aami st79:2017, comprehensive guide to steam sterilization and sterility assurance in health. This article delves into the regulatory standards set for sterile instrument storage and provides insights on how healthcare institutions can effectively meet these standards. Regardless of the location, policies and procedures for sterile storage should be developed according to accepted professional practices to ensure that the function and sterility of medical and surgical devices and supplies are maintained until the time of use. This article provides an overview of the guideline and discusses recommendations for selection of a processing method, transport and storage of sterile items, steam sterilization, and quality.
Comments are closed.