Stem Cell Therapy For Multiple Sclerosis A Pipeline Analysis

Stem Cell Therapy For Multiple Sclerosis A Pipeline Analysis We provide a detailed analysis of the latest developments in preclinical and clinical studies, emphasizing the efficacy and safety of different stem cell types in ms treatment. Mesenchymal stem cell (msc) therapy has emerged as a promising approach due to its immunomodulatory and neuroprotective properties. this systematic review aims to evaluate the therapeutic potential of mesenchymal stem cell (msc) therapy in the treatment of multiple sclerosis (ms).

Stem Cell Therapy For Multiple Sclerosis A Pipeline Analysis Although mesenchymal stem cells (mscs) have been identified as a potential novel therapeutic strategy for multiple sclerosis (ms), existing evidence regarding the effectiveness and safety of this strategy remains inconclusive. The report entails an in depth analysis of the pipeline landscape along with the pipeline molecules in different developmental stages. it also lists out the major regulatory authorities in this market. In this comprehensive review, we provide an outline of current ms medications in the pipeline including emerging dmts and stem cell therapy, as well as the unique characteristics of these medications, including their indications, pharmacokinetic effects, and the relevant advancements. Pipeline adsc for the treatment of multiple sclerosis (ms) clinical trial: a prospective, single center, open label, dose escalation phase i iia study to assess the safety and efficacy of an intrathecal administration of scm 010 in subjects with secondary progressive multiple sclerosis (spms).
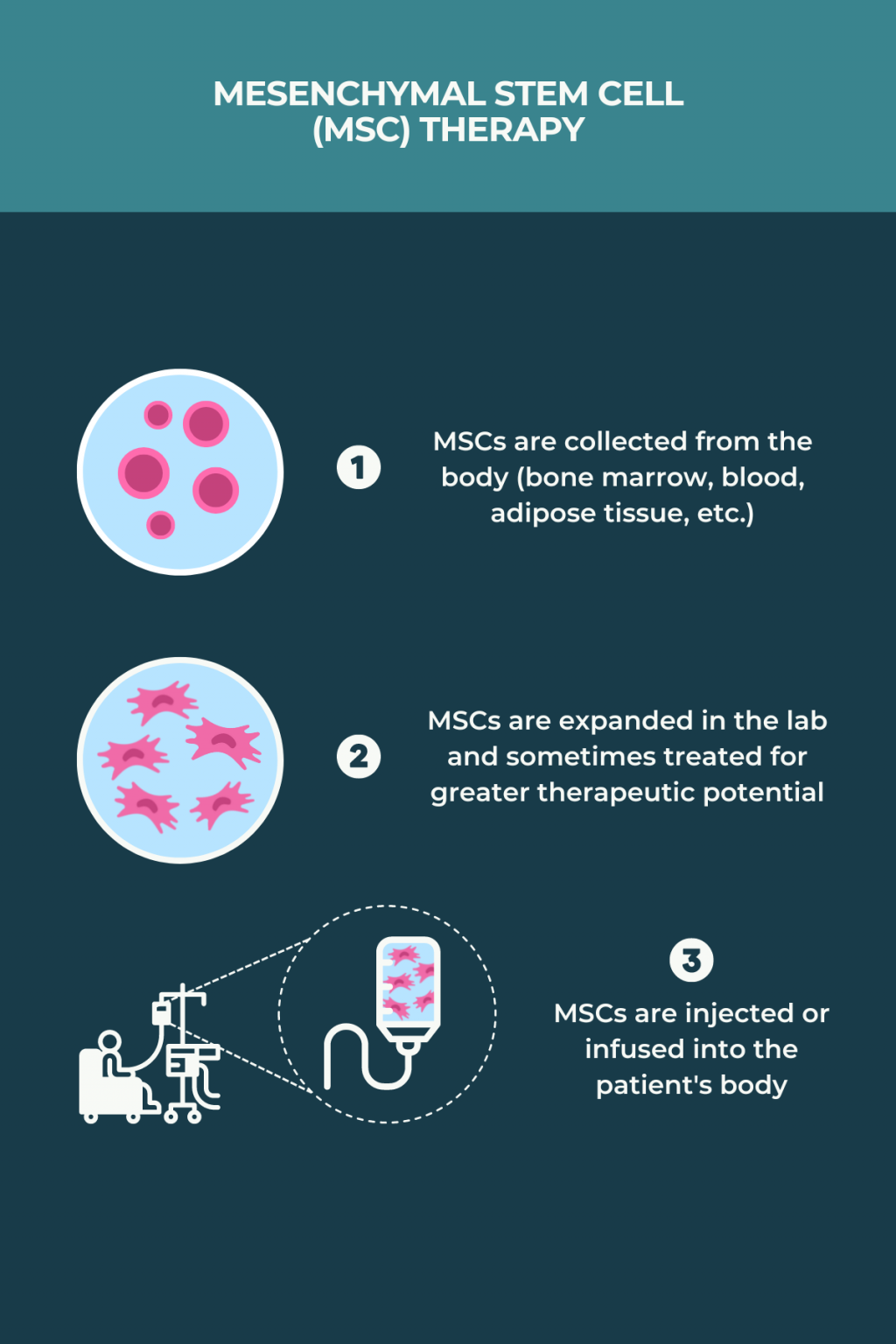
Stem Cell Therapy For Ms Multiple Sclerosis News Today In this comprehensive review, we provide an outline of current ms medications in the pipeline including emerging dmts and stem cell therapy, as well as the unique characteristics of these medications, including their indications, pharmacokinetic effects, and the relevant advancements. Pipeline adsc for the treatment of multiple sclerosis (ms) clinical trial: a prospective, single center, open label, dose escalation phase i iia study to assess the safety and efficacy of an intrathecal administration of scm 010 in subjects with secondary progressive multiple sclerosis (spms). This is a randomised, double blind crossover study to study the effect of intravenous treatment with autologous (derived from the individuals themselves) mesenchymal stem cells (mscs) in patients with multiple sclerosis (ms). current treatments for ms target the immune system and are not curative. Regardless of different individual studies, our meta analysis provides a comprehensive overview showing the potential of msc therapy as a possible effective treatment strategy for patients. To obtain an overview of the therapeutic efficacy of stem cell free therapy in ms, we systematically reviewed preclinical studies of ms treating with stem cell derived cm or evs. This section covers the detailed analysis of each drug under multiple phases, including phase i, phase ii, phase iii, phase iv, and emerging drugs for multiple sclerosis.
Stem Cell Approaches For Multiple Sclerosis Treatment This is a randomised, double blind crossover study to study the effect of intravenous treatment with autologous (derived from the individuals themselves) mesenchymal stem cells (mscs) in patients with multiple sclerosis (ms). current treatments for ms target the immune system and are not curative. Regardless of different individual studies, our meta analysis provides a comprehensive overview showing the potential of msc therapy as a possible effective treatment strategy for patients. To obtain an overview of the therapeutic efficacy of stem cell free therapy in ms, we systematically reviewed preclinical studies of ms treating with stem cell derived cm or evs. This section covers the detailed analysis of each drug under multiple phases, including phase i, phase ii, phase iii, phase iv, and emerging drugs for multiple sclerosis.

Stem Cell Based Therapies For Multiple Sclerosis National Stem Cell To obtain an overview of the therapeutic efficacy of stem cell free therapy in ms, we systematically reviewed preclinical studies of ms treating with stem cell derived cm or evs. This section covers the detailed analysis of each drug under multiple phases, including phase i, phase ii, phase iii, phase iv, and emerging drugs for multiple sclerosis.
Comments are closed.