Specific Heat Equation Stated Clearly
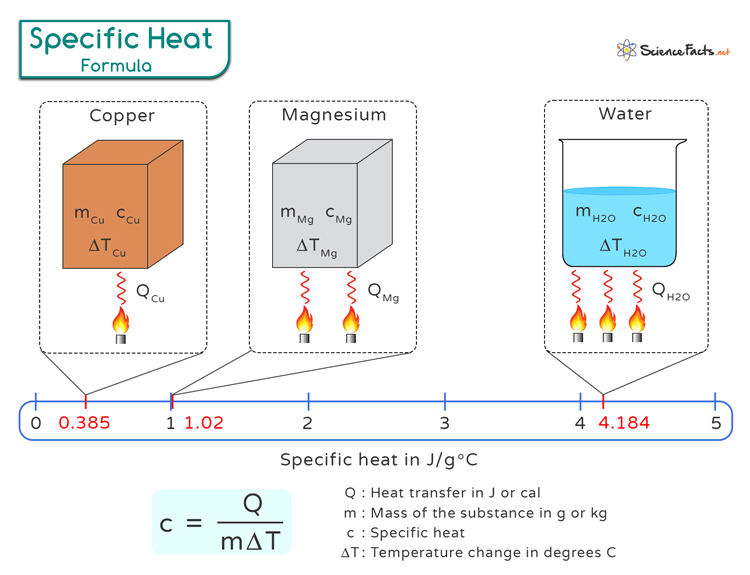
Specific Heat Equation Learn how to use a triangle to memorize and learn how to apply the specific heat equation. The specific heat of a substance can be used to calculate the temperature change that a given substance will undergo when it is either heated or cooled. the equation that relates heat (q) to specific heat (c p), mass (m), and temperature change (Δ t) is shown below. q = c p × m × Δ t.

Specific Heat Equation What is specific heat? learn heat capacity with equations and units, and check out a few specific heat values and example problems. The equation used to calculate specific heat capacity (c) is: c=q mΔt. where: Δt is the change in temperature (in celsius or kelvin). this formula is derived from the relationship between thermal energy and temperature change, emphasizing how the specific heat capacity moderates this relationship. The specific heat of a substance can be used to calculate the temperature change that a given substance will undergo when it is either heated or cooled. the equation that relates heat (q) to specific heat (c p), mass (m), and temperature change (Δ t) is shown below. Specific heat is closely related to the concept of heat capacity. heat capacity is the amount of heat necessary to change the temperature of a substance by 1.00 °c °c . in equation form, heat capacity c is c = m c c = m c, where m is mass and c is specific heat.

Specific Heat Equation The specific heat of a substance can be used to calculate the temperature change that a given substance will undergo when it is either heated or cooled. the equation that relates heat (q) to specific heat (c p), mass (m), and temperature change (Δ t) is shown below. Specific heat is closely related to the concept of heat capacity. heat capacity is the amount of heat necessary to change the temperature of a substance by 1.00 °c °c . in equation form, heat capacity c is c = m c c = m c, where m is mass and c is specific heat. The specific heat equation (q = mcΔt) calculates heat energy transferred during temperature changes. it's essential for calorimetry, thermodynamics, and understanding how substances store thermal energy. In this chemistry tutorial, you will be introduced to the topic of specific heat. in addition, you will learn the formula that goes along with this concept and go through an example to work out the math. There are a few ways to find the specific heat capacity of a material or system, such as the thermal energy equation, the law of dulong and petit, or the einstein debye model. Master the specific heat capacity equation and its role in determining the heat energy required to change the temperature of materials.
Comments are closed.