Solved The Mechanisms Sn1 E1 Sn2 E2 Chegg

Solved For Which Reaction Mechanisms Sn1 Sn2 E1 E2 Is Chegg An exam gives you a substrate, a nucleophile base, and a solvent, and you're supposed to predict whether the reaction runs sn1, sn2, e1, or e2. sometimes it's a mix. sometimes there's no reaction at all. there are four things to check, in order: substrate, nucleophile base, solvent, and temperature. If you haven’t seen my videos on the sn1, sn2, e1, and e2 mechanisms, i strongly encourage you to first watch those and then come back to this video. so, let’s start by a quick review of the important features of each mechanism.

Solved Consider The E1 E2 Sn2 And Sn1 Mechanisms Of The Chegg Tertiary alkyl halides e2 elimination occurs when a base is used, but s n 1 substitution and e1 elimination occur together under neutral conditions, such as in pure ethanol or water. It is relatively easy to separate s n 2 and e2 pathways from s n 1 e1, since both s n 2 and e2 require strong nucleophile or strong base that are usually negatively charged species, while s n 1 e1 require neutral conditions. In these practice problems, we will discuss how to determine if the mechanism is it sn1 sn2 e1 or e2 based on the alkyl and the nucleophile and the base. The main difference between sn1, sn2, e1, and e2 reactions lies in their reaction type (substitution vs elimination), molecularity (uni or bimolecular), mechanism steps (single or multiple), and reagent type (nucleophile vs base).
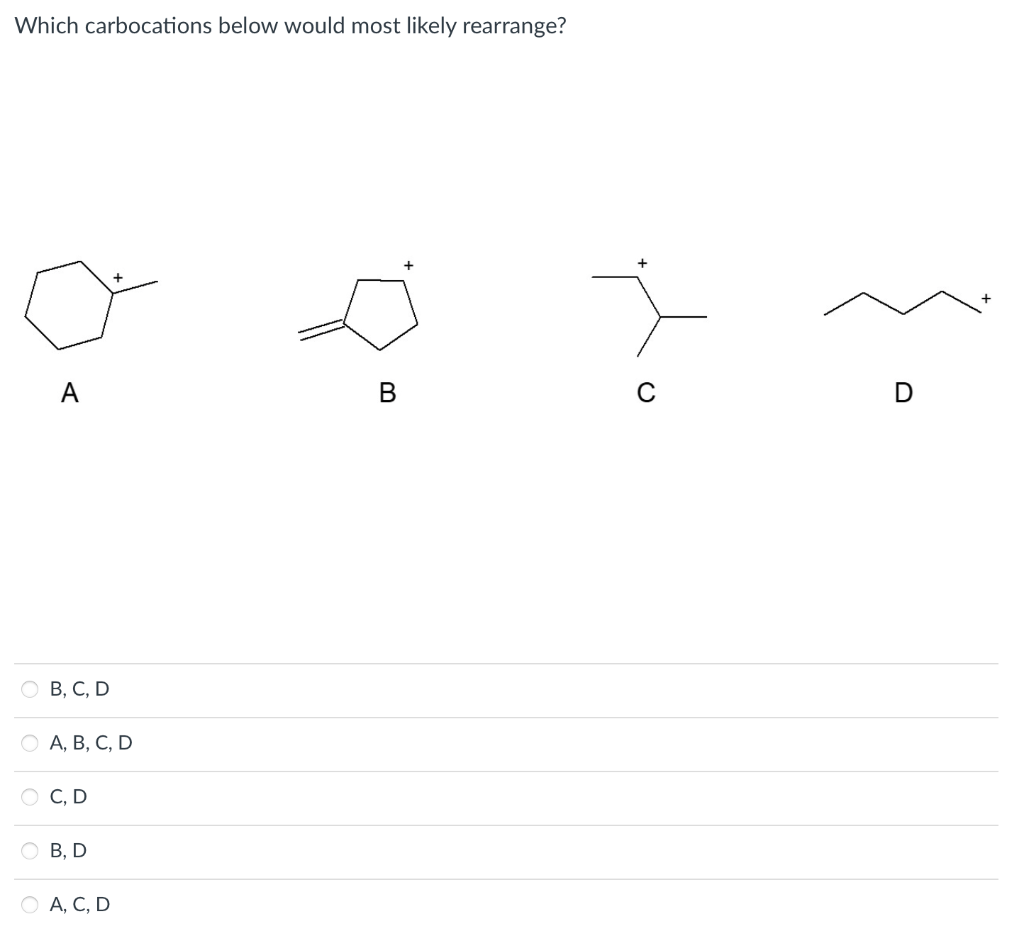
Solved Consider The E1 E2 Sn2 And Sn1 Mechanisms Of The Chegg In these practice problems, we will discuss how to determine if the mechanism is it sn1 sn2 e1 or e2 based on the alkyl and the nucleophile and the base. The main difference between sn1, sn2, e1, and e2 reactions lies in their reaction type (substitution vs elimination), molecularity (uni or bimolecular), mechanism steps (single or multiple), and reagent type (nucleophile vs base). Comprehensive organic chemistry study guide covering sn1, sn2, e1, and e2 mechanisms, carbocation stability, leaving groups, and zaitsev’s rule. Explore the mechanisms of sn1, e1, sn2, and e2 reactions, focusing on key features and factors influencing reaction rates in organic chemistry. Question: for which reaction mechanisms sn1,sn2, e1, or e2 area each of the following statements true? a statement may be true for one or more mechanisms. a. the mechanism involves carbocation intermediates. b. the mechanism has two steps. c. the reaction rate increases with better leaving groups. d. The first set of reactions and mechanisms that are commonly taught are the substitution and elimination reactions. each of these can go by either a one step (sn2 or e2) or two step mechanism (sn1 or e1).
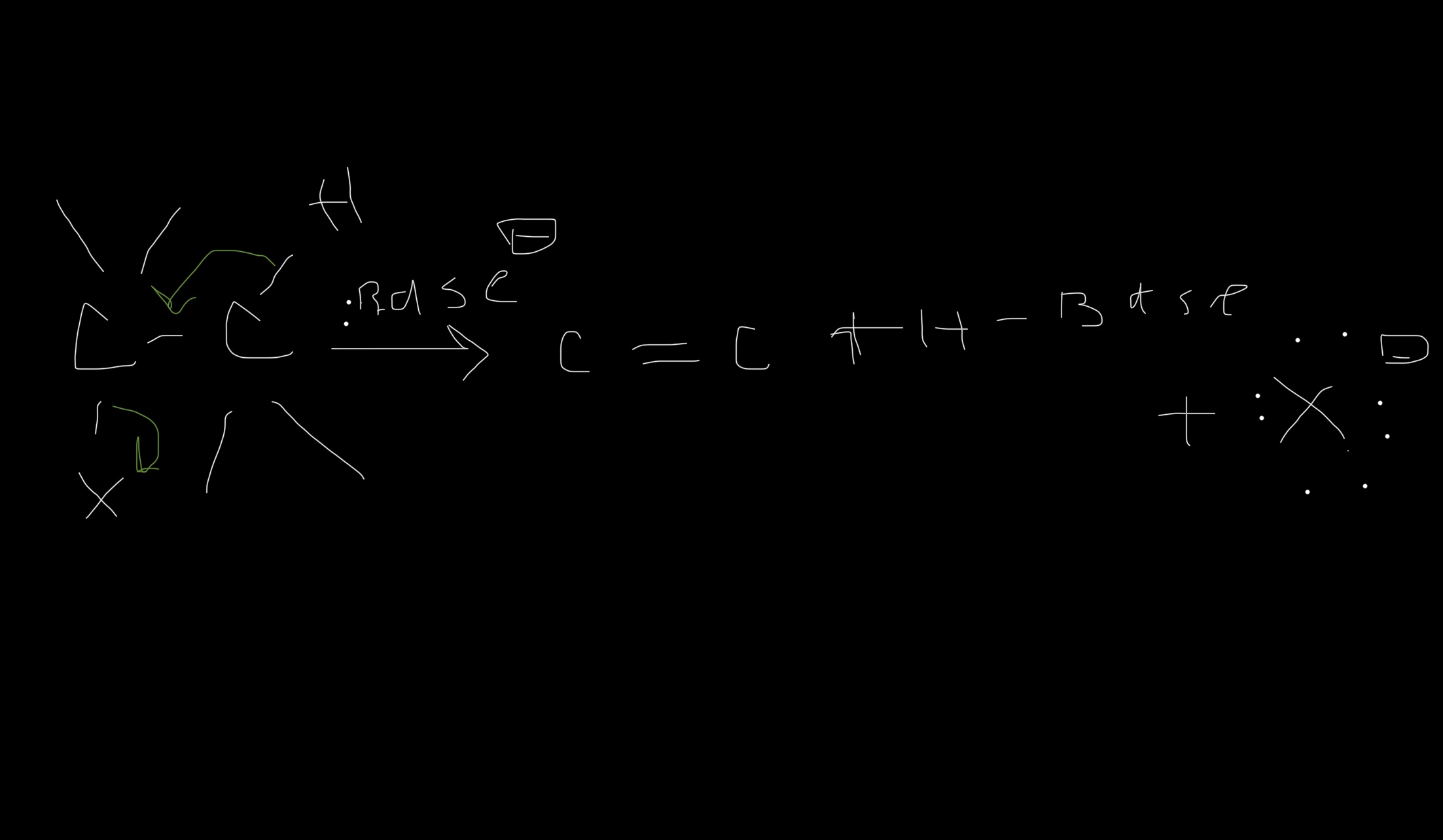
Solved The Mechanisms Sn1 E1 Sn2 E2 Chegg Comprehensive organic chemistry study guide covering sn1, sn2, e1, and e2 mechanisms, carbocation stability, leaving groups, and zaitsev’s rule. Explore the mechanisms of sn1, e1, sn2, and e2 reactions, focusing on key features and factors influencing reaction rates in organic chemistry. Question: for which reaction mechanisms sn1,sn2, e1, or e2 area each of the following statements true? a statement may be true for one or more mechanisms. a. the mechanism involves carbocation intermediates. b. the mechanism has two steps. c. the reaction rate increases with better leaving groups. d. The first set of reactions and mechanisms that are commonly taught are the substitution and elimination reactions. each of these can go by either a one step (sn2 or e2) or two step mechanism (sn1 or e1).
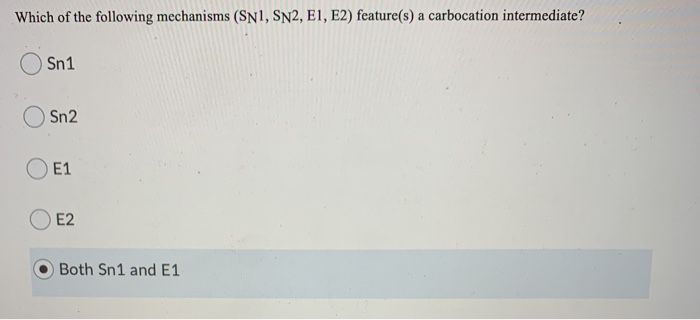
Solved Which Of The Following Mechanisms Sn1 Sn2 E1 E2 Chegg Question: for which reaction mechanisms sn1,sn2, e1, or e2 area each of the following statements true? a statement may be true for one or more mechanisms. a. the mechanism involves carbocation intermediates. b. the mechanism has two steps. c. the reaction rate increases with better leaving groups. d. The first set of reactions and mechanisms that are commonly taught are the substitution and elimination reactions. each of these can go by either a one step (sn2 or e2) or two step mechanism (sn1 or e1).
Comments are closed.