Solved In The Following Reactions Carried Out In Liquid Nh3 Chegg
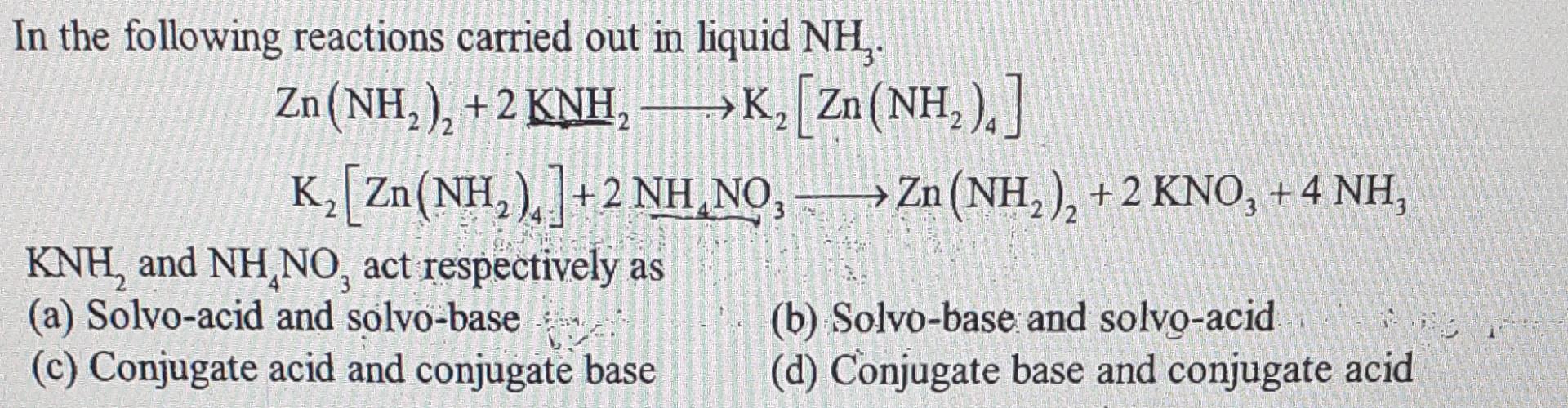
Solved In The Following Reactions Carried Out In Liquid Nh3 Chegg Identify the chemical properties of k n h 2 by evaluating its anionic component, n h 2, which has a 1 charge. the basic property of knh2 is due to the anionic part i.e., nh2 (having 1 c … in the following reactions carried out in liquid nh3. Much of the chemistry in liquid ammonia can be classified by analogy with related reactions in aqueous solutions.

Solved 20 ï Points ï The Following Reaction Is To Be Carried Chegg To do this, we can use the mole ratio between nh3 and n2 in the balanced equation: 4nh3 (l) n2 (g) ¡ú 3n2h4 (l) from this equation, we can see that for every 1 mole of n2, we need 4 moles of nh3. Previous year questions 1 4 of 4 with solutions & explanations on reactions in liquid nh3 (non aqueous solvents) | ias (cse) mains chemistry. answers & detailed explanations available on subscription. Just like water, liquid ammonia undergoes self ionization to form an ammonium ion and amide ion. when sodium metal is added to the liquid ammonia in small amounts, that is, when a dilute solution of sodium in liquid ammonia is formed, we obtain a blue coloured solution. 2.in the following reactions carried out in liquid nh3. zn (nh2)2 2knh2 → k2 [zn ( nh2)4] k2 [zn ( nh2)4] 2nh4no3 → zn (nh2)2 2kno3 4nh3 knh2 and.
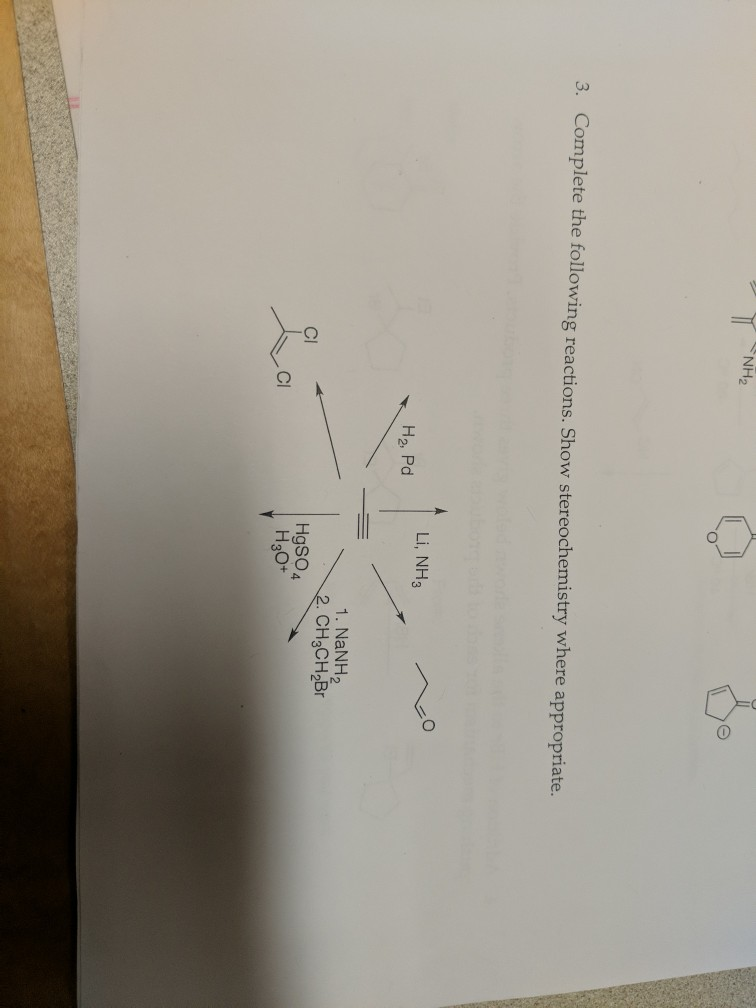
Solved Nh2 3 Complete The Following Reactions Show Chegg Just like water, liquid ammonia undergoes self ionization to form an ammonium ion and amide ion. when sodium metal is added to the liquid ammonia in small amounts, that is, when a dilute solution of sodium in liquid ammonia is formed, we obtain a blue coloured solution. 2.in the following reactions carried out in liquid nh3. zn (nh2)2 2knh2 → k2 [zn ( nh2)4] k2 [zn ( nh2)4] 2nh4no3 → zn (nh2)2 2kno3 4nh3 knh2 and. Question 9. [net june 2015] water plays different roles in the following reactions (i) 2h 2 o ca → ca 2 2oh h 2 (ii) nh 2 o cl → [cl (h 2 o) n] (iii) 6 h 2 o mg 2 → [mg (h 2 o)6] 2. Find step by step chemistry solutions and your answer to the following textbook question: chemists sometimes carry out reactions in liquid ammonia as a solvent.
Comments are closed.