Solution Stoichiometry

Solution Stoichiometry Chemistry Libretexts 59 Off Learn how to calculate the amounts of reactants or products in aqueous solutions using molarity and stoichiometry. see examples of double replacement reactions and concept maps for problem solving. This chemistry video tutorial explains how to solve solution stoichiometry problems.
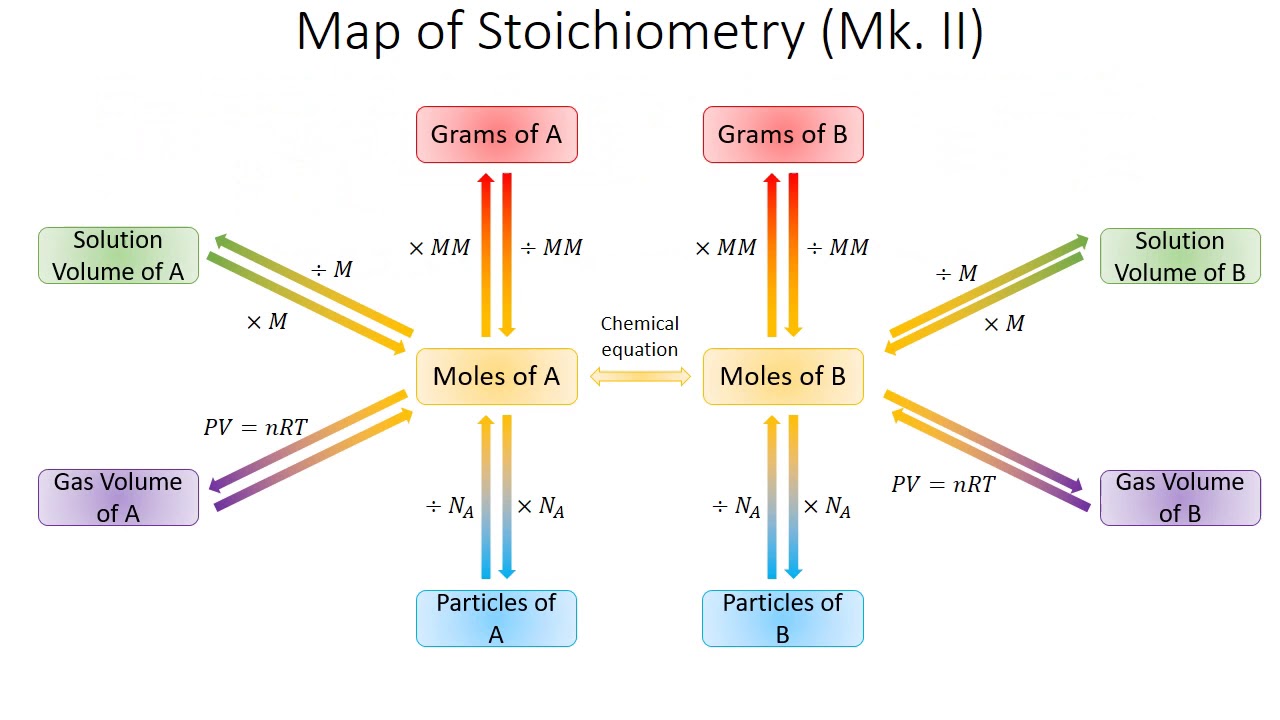
Solution Stoichiometry Chemistry Libretexts 59 Off Learn solution stoichiometry: calculate moles, concentrations, volumes, and conversions in aqueous reactions. step by step examples for chemistry students. Knowing the volume and concentration of a solution containing one reactant, we can determine how much of another solution of another reactant will be needed using the balanced chemical equation. Find out how many moles you need in the solution you’re making. find out what volume of the stock you need for that many moles. Performing stoichiometric calculations involving reactions that occur in solution is referred to as "solution stoichiometry". solution stoichiometry problems, like all stoichiometry problems, requires the use of a mole ratio from a balanced chemical equation.

Solution Stoichiometry Molarity Find out how many moles you need in the solution you’re making. find out what volume of the stock you need for that many moles. Performing stoichiometric calculations involving reactions that occur in solution is referred to as "solution stoichiometry". solution stoichiometry problems, like all stoichiometry problems, requires the use of a mole ratio from a balanced chemical equation. In this section, you'll learn how to calculate molarity, perform dilutions, and use volume and concentration in stoichiometric problem solving. a solution is a homogeneous mixture, meaning its composition is uniform throughout. If given a molar concentration and the solution volume, the number of moles of solute is easy to calculate. once the moles have been determined, we can then use the balanced chemical equation to solve stoichiometry problems that involve solutions. Learn how to calculate molarity and other concentration units of solutions, and how to apply them to chemical reactions. see examples, definitions, and practice problems on solution stoichiometry. Solution stoichiometry is the study of quantitative relationships in chemical reactions that occur in solution. it combines the principles of stoichiometry with solution concentrations to calculate the amounts of reactants and products in reactions involving dissolved substances.

Solution Stoichiometry Overview Examples Expii In this section, you'll learn how to calculate molarity, perform dilutions, and use volume and concentration in stoichiometric problem solving. a solution is a homogeneous mixture, meaning its composition is uniform throughout. If given a molar concentration and the solution volume, the number of moles of solute is easy to calculate. once the moles have been determined, we can then use the balanced chemical equation to solve stoichiometry problems that involve solutions. Learn how to calculate molarity and other concentration units of solutions, and how to apply them to chemical reactions. see examples, definitions, and practice problems on solution stoichiometry. Solution stoichiometry is the study of quantitative relationships in chemical reactions that occur in solution. it combines the principles of stoichiometry with solution concentrations to calculate the amounts of reactants and products in reactions involving dissolved substances.
Comments are closed.