Solution Specific Heat By Cooling Graphical Method Lab Experiment

Solution Specific Heat By Cooling Graphical Method Lab Experiment This experiment aims to determine the specific heat of a liquid using newton's cooling method. the liquid and water are heated to 70 75°c and their cooling rates measured over 25 minutes as they are placed in a double walled chamber. Theory: newton’s law of cooling can be used to determine the specific heat of a liquid by observing the time taken by the liquid in cooling from one temperature to another.

Specific Heat Experiment Cider House Tech This report will be able to help us determine the specific heat of a liquid by the method of cooling.the specific heat of a material is usually known as the amount of heat, needed to increase the temperature of unit mass of a material by 1°c. Sir james joule (1818 1889) studied how water can be warmed by vigorous stirring with a paddle wheel (fig. 17. 15a). the paddle wheel adds energy to the water by doing work on it, and joule found that the temperature rise is directly proportional to the amount of work done. In this experiment, you will learn to determine specific heat capacity of a liquid using newton’s law of cooling. this law states that the rate of loss of heat is directly proportional to the temperature difference between the body and its surroundings, provided it is less than 40qc. This repository contains basic and essential graphs that support various physics lab experiments. these graphs assist in performing calculations, understanding concepts, and analyzing experimental results.

Solution Specific Heat Of Water Experiment Manual Studypool In this experiment, you will learn to determine specific heat capacity of a liquid using newton’s law of cooling. this law states that the rate of loss of heat is directly proportional to the temperature difference between the body and its surroundings, provided it is less than 40qc. This repository contains basic and essential graphs that support various physics lab experiments. these graphs assist in performing calculations, understanding concepts, and analyzing experimental results. Name of the experiment:to determine the specific heat of a liquid by the method of cooling 1.1 objectives: the main objective of this experiment is to determine the specific heat of a given liquid by newton’s cooling method. 1.2 pre lab: student should read the lab manual and have clear idea about the objective, time frame and outcomes of the. Graph two cooling curves in excel – the first for part i when only water was cooling, and the second for part ii when the water and metal specimens were cooling. In this experiment, students will find the specific heat of three “unknown” samples of metal. to do this, students will use their knowledge of thermodynamics to construct a calorimeter. To study the relationship between the temperature of a hot body andnewton’s law of cooling apparatus that includes a copper calorimeter.

Experiment Booklet Heating And Cooling Water Teaching Resources Name of the experiment:to determine the specific heat of a liquid by the method of cooling 1.1 objectives: the main objective of this experiment is to determine the specific heat of a given liquid by newton’s cooling method. 1.2 pre lab: student should read the lab manual and have clear idea about the objective, time frame and outcomes of the. Graph two cooling curves in excel – the first for part i when only water was cooling, and the second for part ii when the water and metal specimens were cooling. In this experiment, students will find the specific heat of three “unknown” samples of metal. to do this, students will use their knowledge of thermodynamics to construct a calorimeter. To study the relationship between the temperature of a hot body andnewton’s law of cooling apparatus that includes a copper calorimeter.
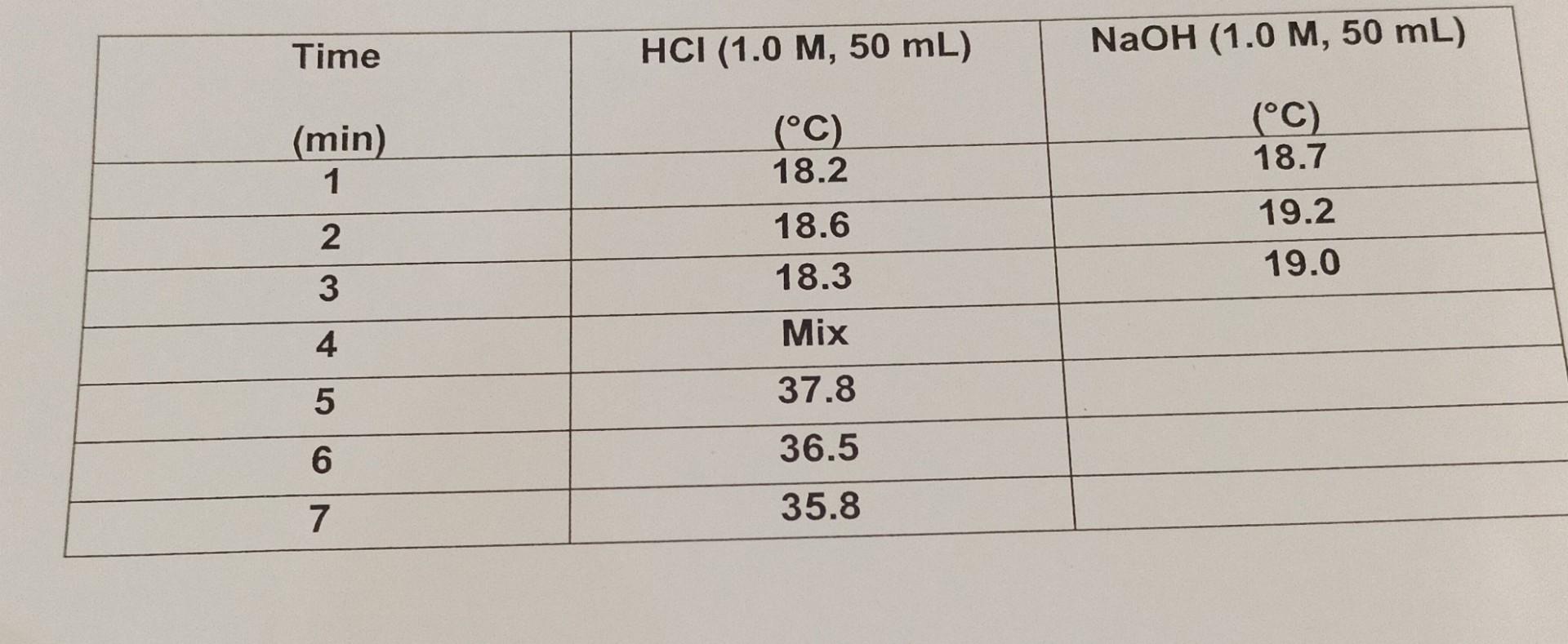
Solved C Determine The Heat Gained By Solution Specific Chegg In this experiment, students will find the specific heat of three “unknown” samples of metal. to do this, students will use their knowledge of thermodynamics to construct a calorimeter. To study the relationship between the temperature of a hot body andnewton’s law of cooling apparatus that includes a copper calorimeter.
Comments are closed.