Solubility Meaning

Solubility Pdf Solution Dissolution Chemistry In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. insolubility is the opposite property, the inability of the solute to form such a solution. Solubility defines how much solute can dissolve under specific conditions. solubility rules help quickly predict the dissolving ability of ionic compounds, often summarized in a solubility chart.

Solubility Infographic Infogram Solubility, degree to which a substance dissolves in a solvent to make a solution (usually expressed as grams of solute per litre of solvent). solubility of one fluid (liquid or gas) in another may be complete (totally miscible; e.g., methanol and water) or partial (oil and water dissolve only. Solubility is the ability of a solute to dissolve in a solvent to form a solution. learn about the properties, types, and factors that affect solubility, such as temperature, pressure, and intermolecular forces. Solubility is defined as the ability of a substance to dissolve in a solvent, with factors such as molecular interactions influencing the extent to which substances like sucrose can dissolve in water. Solubility is the maximum concentration of a solute that can dissolve in a solvent at a given temperature. learn how solubility depends on temperature, pressure, and the type of solute and solvent with examples and equations.

Solubility Meaning Solubility Chemistrybytes Solubility is defined as the ability of a substance to dissolve in a solvent, with factors such as molecular interactions influencing the extent to which substances like sucrose can dissolve in water. Solubility is the maximum concentration of a solute that can dissolve in a solvent at a given temperature. learn how solubility depends on temperature, pressure, and the type of solute and solvent with examples and equations. Solubility is defined as the maximum quantity of a substance that can be dissolved in another. it is the maximum amount of solute that can be dissolved in a solvent at equilibrium, which produces a saturated solution. Solubility is the quality of being able to dissolve in another substance, or the degree to which something can dissolve. learn more about solubility with examples, synonyms, and related words from the cambridge dictionary. : the amount of a substance that will dissolve in a given amount of another substance and is typically expressed as the number of parts by weight dissolved by 100 parts of solvent at a specified temperature and pressure or as percent by weight or by volume. Solubility is the property of a substance, known as the solute, that allows it to be dissolved in a solvent. virtually all substances are either partially or completely soluble in a particular solvent.

Solubility Meaning Solubility Chemistrybytes Solubility is defined as the maximum quantity of a substance that can be dissolved in another. it is the maximum amount of solute that can be dissolved in a solvent at equilibrium, which produces a saturated solution. Solubility is the quality of being able to dissolve in another substance, or the degree to which something can dissolve. learn more about solubility with examples, synonyms, and related words from the cambridge dictionary. : the amount of a substance that will dissolve in a given amount of another substance and is typically expressed as the number of parts by weight dissolved by 100 parts of solvent at a specified temperature and pressure or as percent by weight or by volume. Solubility is the property of a substance, known as the solute, that allows it to be dissolved in a solvent. virtually all substances are either partially or completely soluble in a particular solvent.
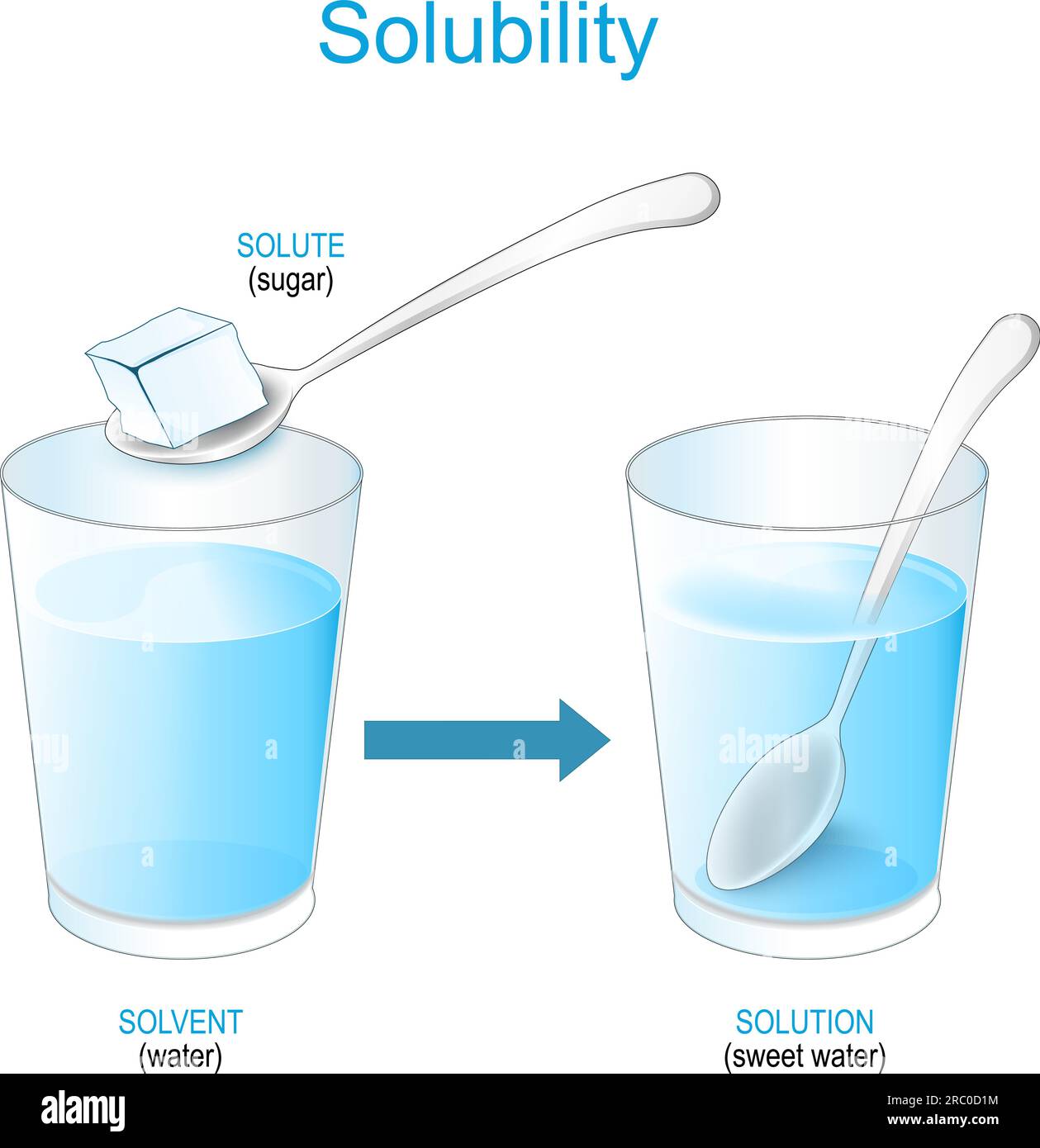
Solubility Definition Javatpoint 47 Off : the amount of a substance that will dissolve in a given amount of another substance and is typically expressed as the number of parts by weight dissolved by 100 parts of solvent at a specified temperature and pressure or as percent by weight or by volume. Solubility is the property of a substance, known as the solute, that allows it to be dissolved in a solvent. virtually all substances are either partially or completely soluble in a particular solvent.
Comments are closed.