Solubility

Solubility General Chemistry Lecture Slides Docsity Solubility is often said to be one of the "characteristic properties of a substance", which means that solubility is commonly used to describe the substance, to indicate a substance's polarity, to help to distinguish it from other substances, and as a guide to applications of the substance. Solubility is defined as the upper limit of solute that can be dissolved in a given amount of solvent at equilibrium. in such an equilibrium, le chatelier's principle can be used to explain most of the main factors that affect solubility.

Dissolving Solids Solubility Chemistry Royalty Free Vector Solubility of one fluid (liquid or gas) in another may be complete (totally miscible; e.g., methanol and water) or partial (oil and water dissolve only slightly). Learn the definition, properties and factors of solubility, the ability of a solute to dissolve in a solvent to form a solution. explore the effects of temperature, pressure, bonds and intermolecular forces on solubility with examples and interactive video. Learn what solubility is, how it is measured, and what factors influence it. see examples of solubility for different types of solutes and solvents, such as salt, sugar, carbon dioxide, and ethylene glycol. Solubility is defined as the ability of a substance to dissolve in a solvent, with factors such as molecular interactions influencing the extent to which substances like sucrose can dissolve in water.
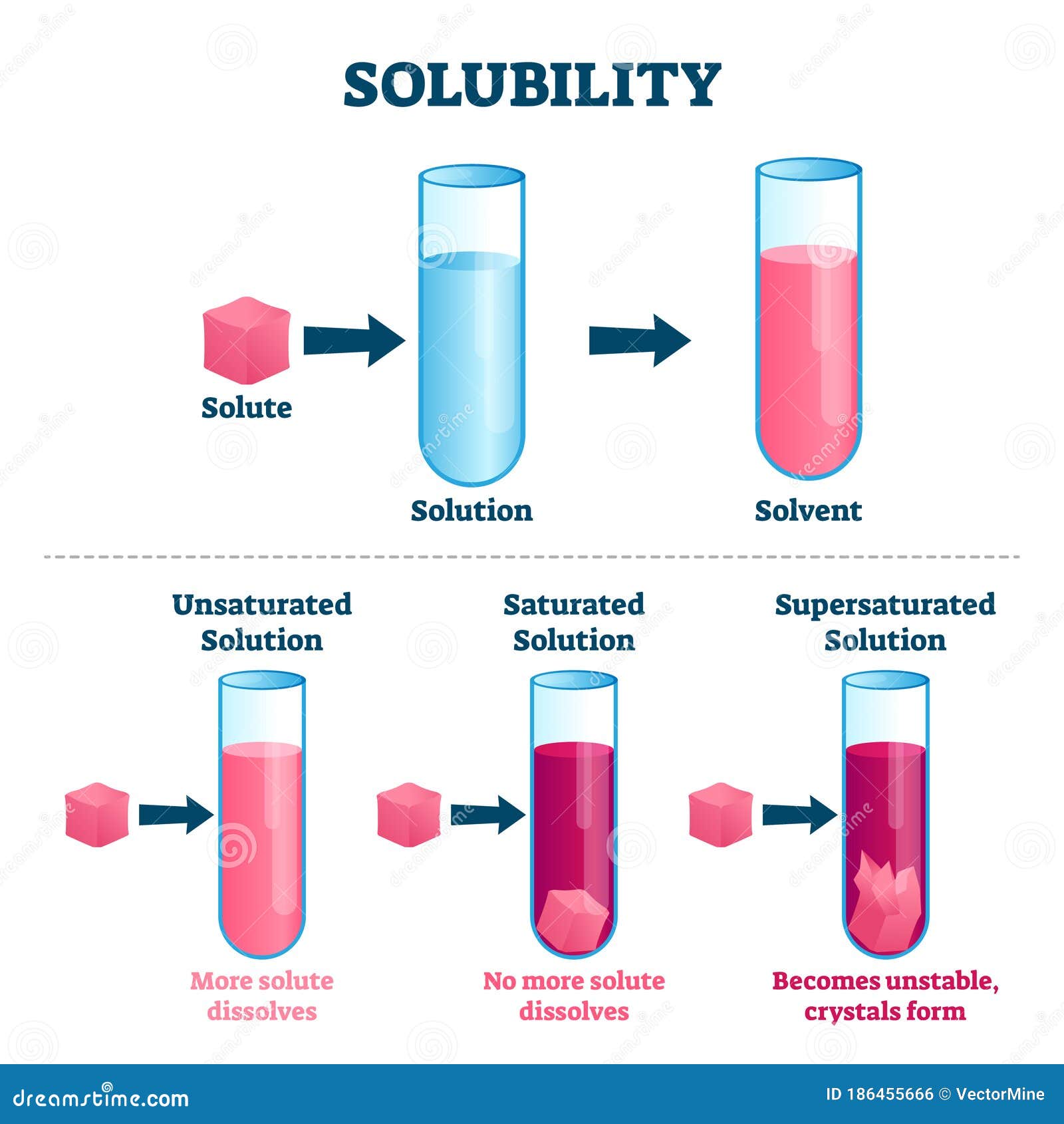
Solubility Vector Illustration Labeled Solute Solvent And Solution Learn what solubility is, how it is measured, and what factors influence it. see examples of solubility for different types of solutes and solvents, such as salt, sugar, carbon dioxide, and ethylene glycol. Solubility is defined as the ability of a substance to dissolve in a solvent, with factors such as molecular interactions influencing the extent to which substances like sucrose can dissolve in water. The solubility of a solute in a particular solvent is the maximum concentration that may be achieved under given conditions when the dissolution process is at equilibrium. Learn about the factors that affect the solubility of solids in water, such as intermolecular forces, ionic bonds, and solubility rules. find out how to predict whether a salt is soluble, insoluble, or slightly soluble in water based on its composition and properties. Discover what solubility means, factors affecting it, and real life examples for easy student understanding. Solubility is defined as the maximum quantity of a substance that can be dissolved in another. it is the maximum amount of solute that can be dissolved in a solvent at equilibrium, which produces a saturated solution.
Comments are closed.