Solubility And Different Liquidssubscribescience Viral Youtubeshorts Shortvideo Shortsshort

Solubility And Different Liquids Subscribe Science Viral Solubility and different liquids subscribe science viral shorts shortvideo #scienceshorts. Enjoy the videos and music you love, upload original content, and share it all with friends, family, and the world on .

Solubility In High And Low Concentration Liquid Solubility Subscribed 12 1.1k views 4 months ago solubility and different liquid||chemistry||science|| #viral #science #foryou #shorts #solubility density in different liquid more. Video transcript [instructor] in this video, we're going to talk about solubility, which is just a way of describing how well certain solutes can dissolve in certain solvents. and just as an example, we could go to our old friend sodium chloride and think about why does it dissolve well in water. The greater the difference in molecular structure (and hence in intermolecular attractions), the lower the mutual solubility. the following video succinctly showcases this principle. In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. insolubility is the opposite property, the inability of the solute to form such a solution.
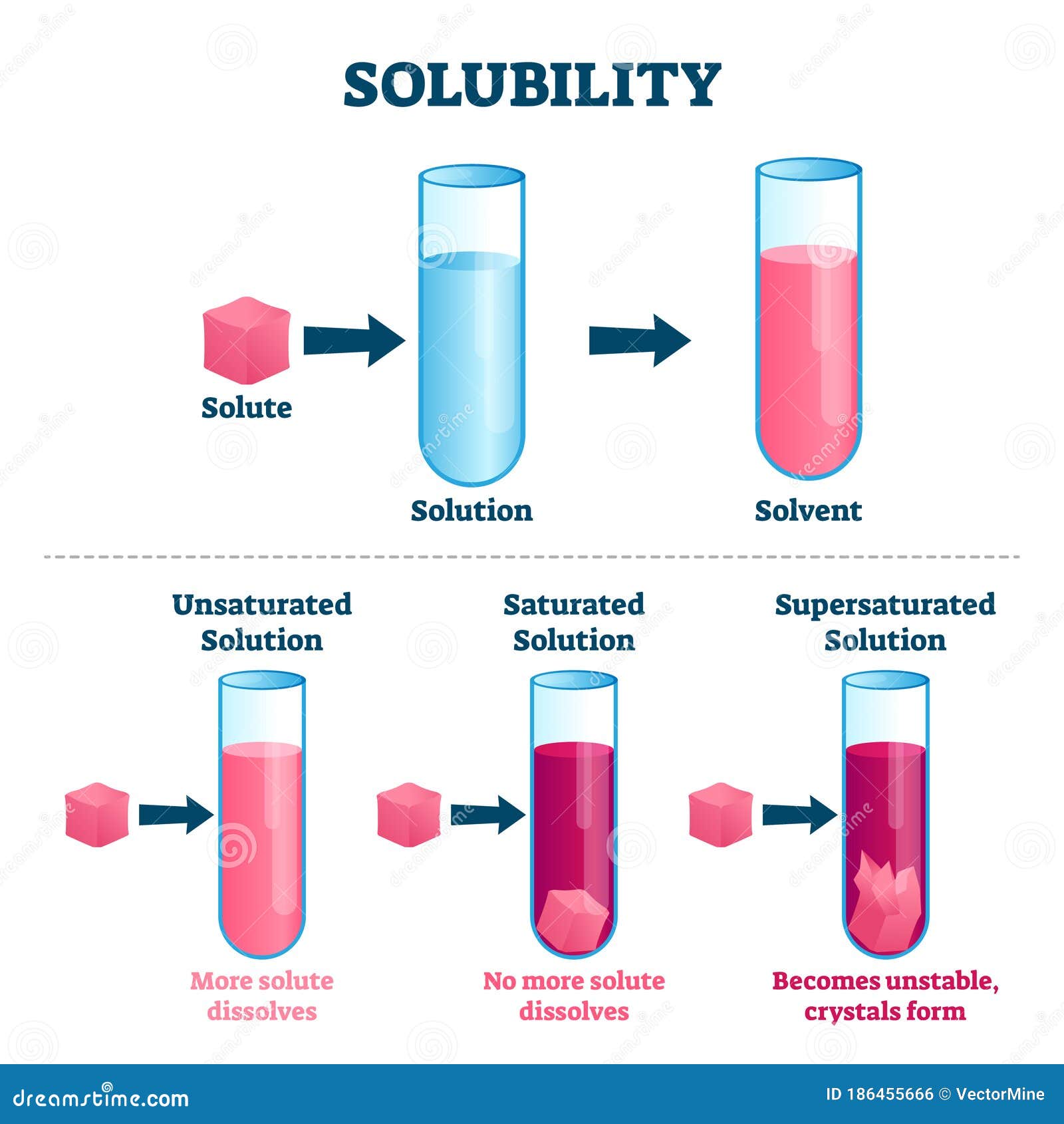
Solubility In Water Animation The greater the difference in molecular structure (and hence in intermolecular attractions), the lower the mutual solubility. the following video succinctly showcases this principle. In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. insolubility is the opposite property, the inability of the solute to form such a solution. Add different salts to water, then watch them dissolve and achieve a dynamic equilibrium with solid precipitate. compare the number of ions in solution for highly soluble nacl to other slightly soluble salts. Solubility is the ability of a solid, liquid, or gaseous chemical substance (referred to as the solute) to dissolve in solvent (usually a liquid) and form a solution. the solubility of a substance fundamentally depends on the solvent used, as well as temperature and pressure. Explore how substances dissolve in water, understand solubility rules, and discover the factors affecting solubility. engage in exciting experiments and a solubility scavenger hunt. uncover the secrets of solubility in this fun and educational science lesson for kindergarteners. Miscible liquids are soluble in all proportions, and immiscible liquids exhibit very low mutual solubility. solubilities for gaseous solutes decrease with increasing temperature, while those for most, but not all, solid solutes increase with temperature.

Solubility Concept Solubility Varies With Temperature Solubility Add different salts to water, then watch them dissolve and achieve a dynamic equilibrium with solid precipitate. compare the number of ions in solution for highly soluble nacl to other slightly soluble salts. Solubility is the ability of a solid, liquid, or gaseous chemical substance (referred to as the solute) to dissolve in solvent (usually a liquid) and form a solution. the solubility of a substance fundamentally depends on the solvent used, as well as temperature and pressure. Explore how substances dissolve in water, understand solubility rules, and discover the factors affecting solubility. engage in exciting experiments and a solubility scavenger hunt. uncover the secrets of solubility in this fun and educational science lesson for kindergarteners. Miscible liquids are soluble in all proportions, and immiscible liquids exhibit very low mutual solubility. solubilities for gaseous solutes decrease with increasing temperature, while those for most, but not all, solid solutes increase with temperature.
Comments are closed.