Solubility Concept Solubility Varies With Temperature Solubility
Effect Of Temperature On Solubility Qs Study The solubility of the majority of solid substances increases as the temperature increases. however, the effect is difficult to predict and varies widely from one solute to another. The solubility mainly depends on the composition of solute and solvent (including their ph and the presence of other dissolved substances) as well as on temperature and pressure.
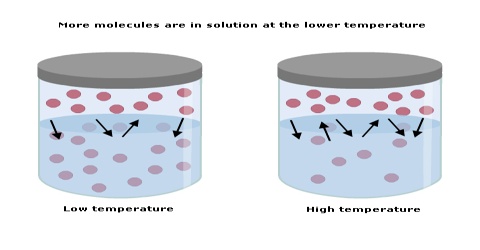
Solved The Graph Below Shows How Solubility Changes With Temperature Solubility is specific for a particular solvent. we will consider solubility of material in water as solvent. the solubility of the majority of solid substances increases as the temperature increases. however, the effect is difficult to predict and varies widely from one solute to another. Temperature is another factor affecting solubility, with gas solubility typically decreasing as temperature increases ((figure)). this inverse relation between temperature and dissolved gas concentration is responsible for one of the major impacts of thermal pollution in natural waters. Solubility, the ability of a substance to dissolve in a solvent, is not a static characteristic but varies with changes in environmental conditions such as temperature. Solubility is specific for a particular solvent. we will consider solubility of material in water as solvent. the solubility of the majority of solid substances increases as the temperature increases. however, the effect is difficult to predict and varies widely from one solute to another.

Predicting How Solubility Varies With Temperature Chemistry Study Solubility, the ability of a substance to dissolve in a solvent, is not a static characteristic but varies with changes in environmental conditions such as temperature. Solubility is specific for a particular solvent. we will consider solubility of material in water as solvent. the solubility of the majority of solid substances increases as the temperature increases. however, the effect is difficult to predict and varies widely from one solute to another. Learn how to predict how solubility varies with temperature, and see examples that walk through sample problems step by step for you to improve your chemistry knowledge and skills. Temperature is another factor affecting solubility, with gas solubility typically decreasing as temperature increases ((figure)). this inverse relation between temperature and dissolved gas concentration is responsible for one of the major impacts of thermal pollution in natural waters. The dependence of solubility on temperature for a number of solids in water is shown by the solubility curves in figure 11.16. reviewing these data indicates a general trend of increasing solubility with temperature, although there are exceptions, as illustrated by the ionic compound cerium sulfate. There appear to be strange limits to this solubility: depending on the solution, raising or lowering the temperature can result in phase separation!.

Of A Substance Varies With A Temperature Solubility Is Maximum Amount Of Learn how to predict how solubility varies with temperature, and see examples that walk through sample problems step by step for you to improve your chemistry knowledge and skills. Temperature is another factor affecting solubility, with gas solubility typically decreasing as temperature increases ((figure)). this inverse relation between temperature and dissolved gas concentration is responsible for one of the major impacts of thermal pollution in natural waters. The dependence of solubility on temperature for a number of solids in water is shown by the solubility curves in figure 11.16. reviewing these data indicates a general trend of increasing solubility with temperature, although there are exceptions, as illustrated by the ionic compound cerium sulfate. There appear to be strange limits to this solubility: depending on the solution, raising or lowering the temperature can result in phase separation!.
Comments are closed.