Single Stage Batch Distillation Example Part 2

Che 200 Week 12 Batch Distillation Pdf Distillation Chemical Organized by textbook: learncheme demonstrates using an excel spreadsheet to solve the rayleigh equation for a single stage batch distillation in order to determine the amount. Example problem 2 a single stage batch still operating at 760 mm hg pressure is charged with 75 mol of a 82 mol% methanol (1) 18 mol% water (2) mixture. assume the mixture is an ideal solution. how much distillate will be collected if the average distillate concentration desired is 90.0%?.
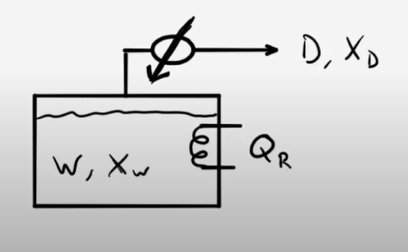
Single Stage Batch Distillation Example Problems Learncheme This document describes the process and equations for simple binary batch distillation. it can be modeled using a rayleigh equation that relates the fraction of material remaining in the still pot to the vapor liquid equilibrium. A mixture of 62 mol% methanol and the remaining water is distilled using a batch distillation. the batch distillation system comprises a still pot and a column with one equilibrium stage. An interactive simulation designed to teach engineering students about single stage batch distillation. Simple distillation, also known as rayleigh distillation or differential distillation , is the most elementary example of batch distillation. in this distillation system, the vapor is removed from the still during a particular time interval and is condensed in the condenser.

Single Stage Batch Distillation Summary Learncheme An interactive simulation designed to teach engineering students about single stage batch distillation. Simple distillation, also known as rayleigh distillation or differential distillation , is the most elementary example of batch distillation. in this distillation system, the vapor is removed from the still during a particular time interval and is condensed in the condenser. Simple binary rayleigh distillation. a simple batch still (one equilibrium stage) is separating a 50 mole feed charge to the still pot that is 80.0 mol% methanol and 20.0 mol% water. an average distillate concentration of 89.2 mol% methanol is required. As the name indicates, this type of distillation is a batch process and not a continuous process. the figure 1 setup consists of a container where a fixed quantity of the liquid mixture is fed initially. A mixture of 62 mol% methanol and the remaining water is distilled using a batch distillation. the batch distillation system comprises a still pot and a column with one equilibrium stage. The simple form of batch distillation involves a single flash separation and is used where a large difference in volatility exists between the components. such as distillation doesn’t need a fractionating column between the batches still, normally a stirred jacketed reactor, and the condenser.

Single Stage Batch Distillation Summary Learncheme Simple binary rayleigh distillation. a simple batch still (one equilibrium stage) is separating a 50 mole feed charge to the still pot that is 80.0 mol% methanol and 20.0 mol% water. an average distillate concentration of 89.2 mol% methanol is required. As the name indicates, this type of distillation is a batch process and not a continuous process. the figure 1 setup consists of a container where a fixed quantity of the liquid mixture is fed initially. A mixture of 62 mol% methanol and the remaining water is distilled using a batch distillation. the batch distillation system comprises a still pot and a column with one equilibrium stage. The simple form of batch distillation involves a single flash separation and is used where a large difference in volatility exists between the components. such as distillation doesn’t need a fractionating column between the batches still, normally a stirred jacketed reactor, and the condenser.
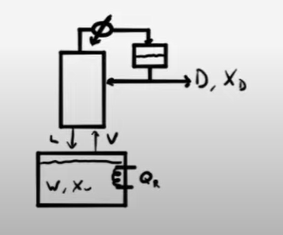
Multi Stage Batch Distillation Example Problems Learncheme A mixture of 62 mol% methanol and the remaining water is distilled using a batch distillation. the batch distillation system comprises a still pot and a column with one equilibrium stage. The simple form of batch distillation involves a single flash separation and is used where a large difference in volatility exists between the components. such as distillation doesn’t need a fractionating column between the batches still, normally a stirred jacketed reactor, and the condenser.
Comments are closed.