Simple Ph Scale

Basic Ph Scale Ph Value Scale Chart Acid Base Balance Infographic Ph is a scale used to measure the acidity or basicity of a solution. it ranges from 0 to 14 and reflects the relative concentration of hydrogen ions (h⁺) and hydroxide ions (oh⁻) in aqueous solutions. The ph scale shows how acidic or basic a chemical is in aqueous solution (mixed with water). the scale runs from 0 (most acidic) to 14 (most alkaline or basic), where 7 is neutral ph.
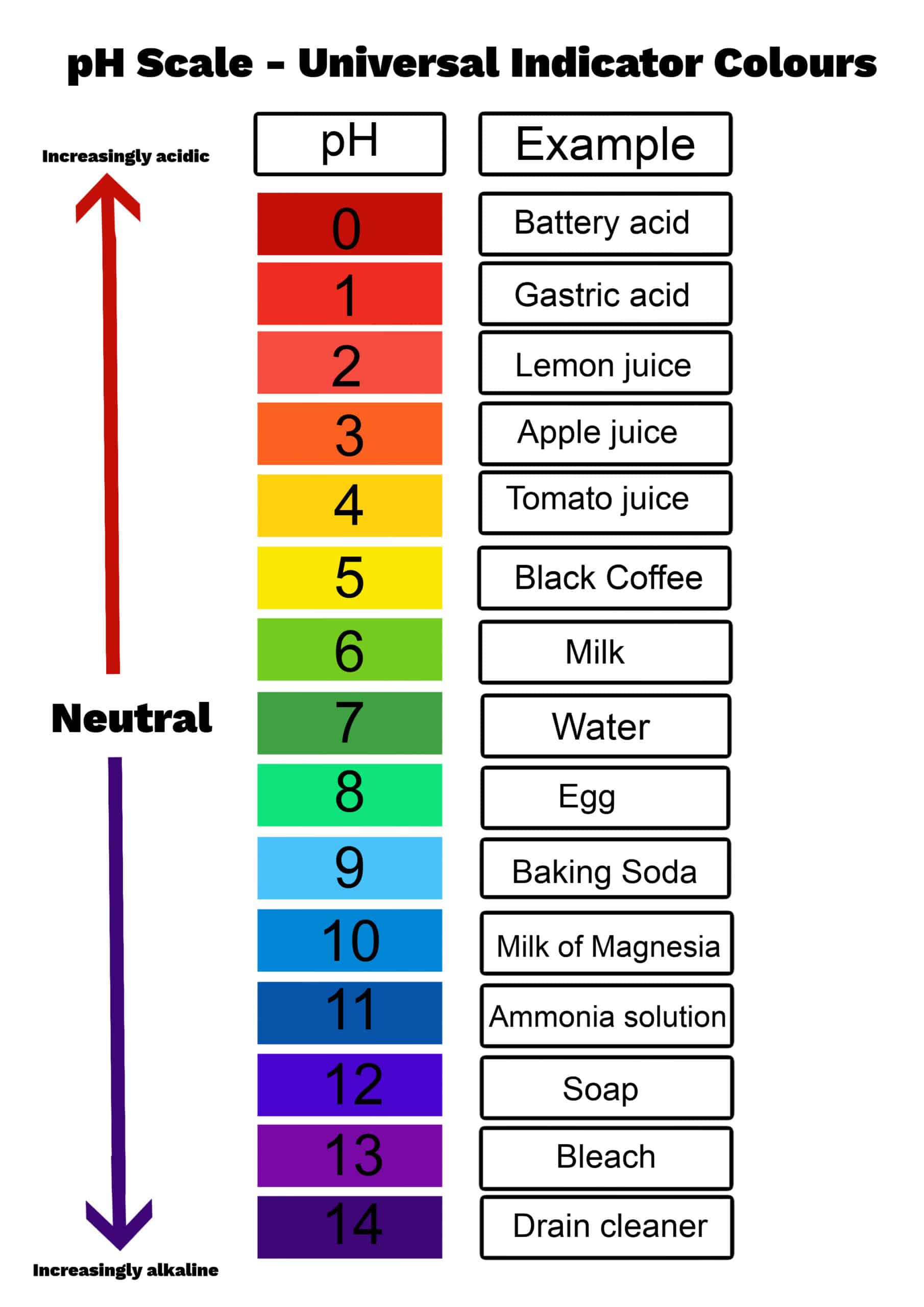
Simple Ph Scale Explore the meaning of ph and how it relates to acidity and basicity. learn how to calculate ph, poh, [h₃o⁺], and [oh⁻], and understand what ph tells you about a solution. Interactive ph scale chart with examples, colors, and detailed explanations. perfect for students and educators learning about acidity and alkalinity. Explore the basics of ph scale with interactive simulations to understand acidity and alkalinity in various solutions. The ph scale measures how acidic or basic (alkaline) a substance is. it ranges from 0 to 14. a ph of 7 is neutral. values below 7 are acidic, and values above 7 are basic. pure water has a neutral ph of 7. when substances dissolve in water, they can shift the ph.

Simple Ph Scale Explore the basics of ph scale with interactive simulations to understand acidity and alkalinity in various solutions. The ph scale measures how acidic or basic (alkaline) a substance is. it ranges from 0 to 14. a ph of 7 is neutral. values below 7 are acidic, and values above 7 are basic. pure water has a neutral ph of 7. when substances dissolve in water, they can shift the ph. Ph is a logarithmic scale. a solution that has a ph of 1.0 has 10 times the [h ] as a solution with a ph of 2.0, which in turn has 10 times the [h ] as a solution with a ph of 3.0 and so forth. In chemistry, ph is used to gauge the acidity or basicity of an aqueous solution. it stands for "potential of hydrogen" and is defined as the negative logarithm of the hydrogen ion (h⁺) concentration. the ph scale values extend from 0 to 14, with a value of seven considered neutral. This is known as the ph scale and is the range of values from 0 to 14 that describes the acidity or basicity of a solution. you can use ph to quickly determine whether a given aqueous solution is acidic, basic, or neutral. The ph scale provides a range of acidic, neutral and basic solutions between 0 14. the ph scale is directly related to the oh concentration while inversely related to the h concentration.

Basic Ph Scale Ph Value Scale Chart Acid Base Balance Infographic Ph is a logarithmic scale. a solution that has a ph of 1.0 has 10 times the [h ] as a solution with a ph of 2.0, which in turn has 10 times the [h ] as a solution with a ph of 3.0 and so forth. In chemistry, ph is used to gauge the acidity or basicity of an aqueous solution. it stands for "potential of hydrogen" and is defined as the negative logarithm of the hydrogen ion (h⁺) concentration. the ph scale values extend from 0 to 14, with a value of seven considered neutral. This is known as the ph scale and is the range of values from 0 to 14 that describes the acidity or basicity of a solution. you can use ph to quickly determine whether a given aqueous solution is acidic, basic, or neutral. The ph scale provides a range of acidic, neutral and basic solutions between 0 14. the ph scale is directly related to the oh concentration while inversely related to the h concentration.
Comments are closed.