Simple Crystallization Process
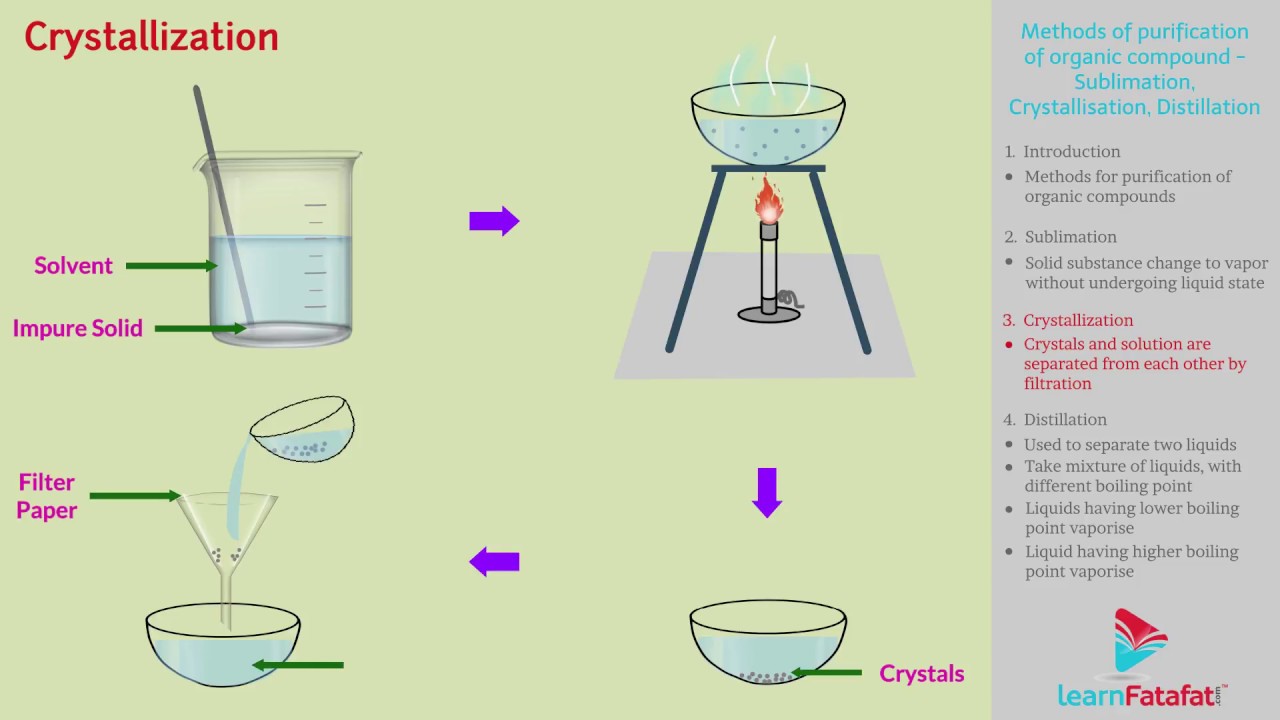
Simple Crystallization Process Crystallization, a fundamental process in nature and industry, is pivotal in forming structured solids from liquids or gases. it involves the orderly arrangement of atoms, ions, or molecules into a highly organized, repeating pattern known as a crystal lattice. Crystallization requires two events to occur. first, on the microscopic scale, atoms or molecules cluster together in a process known as nucleation. if the clusters become stable and large enough, crystal formation may ensue. nucleation is the initial stage or phase in the crystallization process.

Simple Crystallization Process In chemistry, crystallization is commonly used to separate and purify solid substances from a liquid solution. during this process, the dissolved particles organize themselves into a repeating three dimensional pattern called a crystal lattice. Crystallization is a physical process in which a solid forms from a solution, melt, or vapor, creating a highly organized structure called a crystal. this transformation occurs when the solubility conditions change, leading to nucleation and growth of crystal lattices. Crystallization is an excellent purification technique for solids because a crystal slowly forming from a saturated solution tends to selectively incorporate particles of the same type into its crystal structure (modeled below). Learn about crystallization, its processes, types, and real life uses. clear explanations and examples for students.
Simple Crystallization Process Crystallization is an excellent purification technique for solids because a crystal slowly forming from a saturated solution tends to selectively incorporate particles of the same type into its crystal structure (modeled below). Learn about crystallization, its processes, types, and real life uses. clear explanations and examples for students. The crystallization process consists of two major events. one is nucleation and the other is crystal growth which both are driven by thermodynamic properties also as chemical properties. Crystallization occurs in two main phases. the first is nucleation, the appearance of a crystalline phase from either a supercooled liquid or a supersaturated solvent. the second step is known as crystal growth, which is the increase in the size of particles and leads to a crystal state. While the method chosen to crystallize product can vary based on several factors, there are six common steps for crystallization to occur. scientists use solubility curves to create a framework to develop the desired crystallization process. Crystallization can be defined as the process through which the atoms molecules of a substance arrange themselves in a well defined three dimensional lattice and consequently, minimize the overall energy of the system.

Simple Crystallization Process The crystallization process consists of two major events. one is nucleation and the other is crystal growth which both are driven by thermodynamic properties also as chemical properties. Crystallization occurs in two main phases. the first is nucleation, the appearance of a crystalline phase from either a supercooled liquid or a supersaturated solvent. the second step is known as crystal growth, which is the increase in the size of particles and leads to a crystal state. While the method chosen to crystallize product can vary based on several factors, there are six common steps for crystallization to occur. scientists use solubility curves to create a framework to develop the desired crystallization process. Crystallization can be defined as the process through which the atoms molecules of a substance arrange themselves in a well defined three dimensional lattice and consequently, minimize the overall energy of the system.

Simple Crystallization Process While the method chosen to crystallize product can vary based on several factors, there are six common steps for crystallization to occur. scientists use solubility curves to create a framework to develop the desired crystallization process. Crystallization can be defined as the process through which the atoms molecules of a substance arrange themselves in a well defined three dimensional lattice and consequently, minimize the overall energy of the system.

Simple Crystallization Process
Comments are closed.