Silicon Dioxide Glass Structure
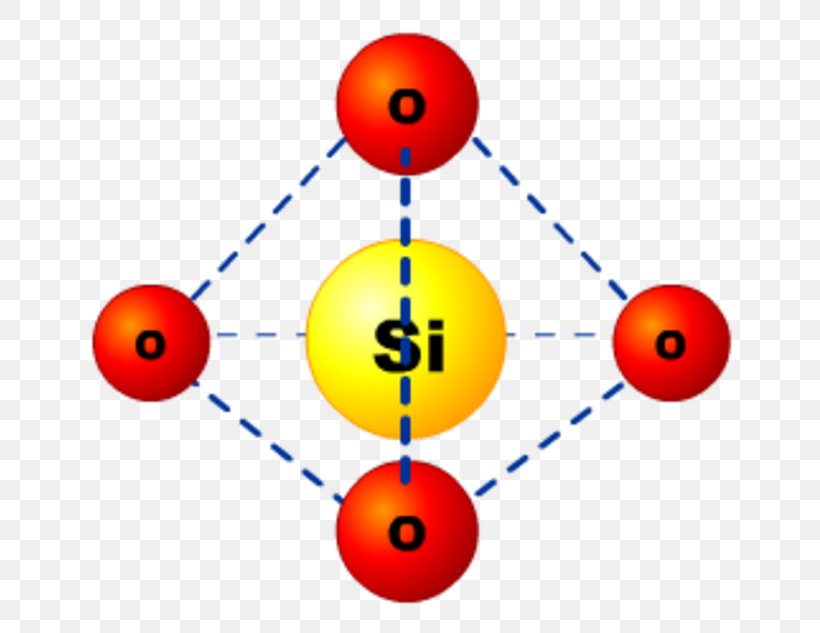
Silicon Dioxide Glass Molecule Structure Png 760x633px Silicon Silicon dioxide is a common fundamental constituent of glass. in the majority of silicon dioxides, the silicon atom shows tetrahedral coordination, with four oxygen atoms surrounding a central si atom (see 3 d unit cell). Found naturally in sand and quartz, it forms when silicon and oxygen atoms bond together in a strong covalent bond, creating a stable and rigid structure. this compound is not only crucial in the manufacturing of glass and ceramics but also in various technological applications.
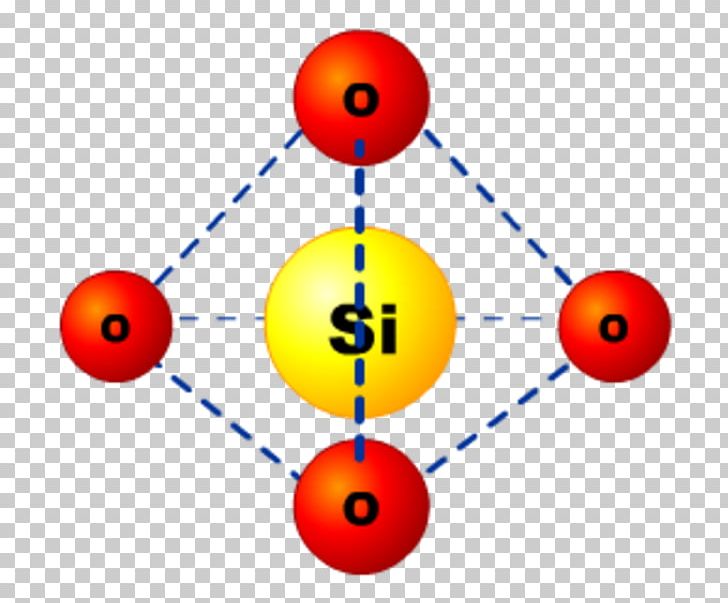
Silicon Dioxide Glass Molecule Structure Png Clipart Area Carbon The physical properties and the structure of noncrystal line silica are of central importance in solid state physics and material sciences because sio is a prototypical glass. Silicon dioxide is used to make glass, while different types of clays are used to make traditional clay ceramics such as porcelain, china and bricks. you may notice that the structure of silicon dioxide or silica is very ordered; with atoms arranged in a regular three dimensional pattern. Glass is an inorganic permanent material, usually made up of silicon dioxide as main structural component, as well as alkaline and earth alkaline metal oxides. because of its molecular structure, glass is an inert material with high barrier properties, hence even small molecules cannot pass through glass. Glass is an amorphous solid, characterized by an ordered yet disordered atomic structure without a crystalline lattice. its composition can vary widely, typically consisting of silica (silicon dioxide) combined with other elements such as boron, lead, or alkali metals.

Silicon Dioxide Glass Structure Glass is an inorganic permanent material, usually made up of silicon dioxide as main structural component, as well as alkaline and earth alkaline metal oxides. because of its molecular structure, glass is an inert material with high barrier properties, hence even small molecules cannot pass through glass. Glass is an amorphous solid, characterized by an ordered yet disordered atomic structure without a crystalline lattice. its composition can vary widely, typically consisting of silica (silicon dioxide) combined with other elements such as boron, lead, or alkali metals. The main constituent of flat glass is sio2 (silica sand). this has a high melting temperature in the region of 1700 degrees c and its state at this temperature is like syrup on a very cold day. The document discusses the fundamentals of glasses, including their definitions, components, and types, with a focus on silicate ceramics and their structures. it explains the manufacturing processes, thermal and mechanical properties, and the significance of various oxides in glass composition. Silicon dioxide, sio a 2, also known as silica is a linear molecule that is formed by one silicon atom and two oxygen atoms with two sets of doubles bonds and 4 single bonds. because of its main component: glass, silicon dioxide is a very common and important molecule in the construction industry. Silicon dioxide, or sio2, has a tetrahedral molecular structure where each silicon atom is bonded to four oxygen atoms. this arrangement forms a strong, stable network found in quartz and glass.
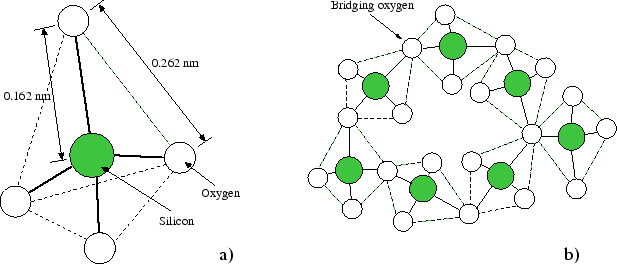
Silicon Dioxide Glass Structure The main constituent of flat glass is sio2 (silica sand). this has a high melting temperature in the region of 1700 degrees c and its state at this temperature is like syrup on a very cold day. The document discusses the fundamentals of glasses, including their definitions, components, and types, with a focus on silicate ceramics and their structures. it explains the manufacturing processes, thermal and mechanical properties, and the significance of various oxides in glass composition. Silicon dioxide, sio a 2, also known as silica is a linear molecule that is formed by one silicon atom and two oxygen atoms with two sets of doubles bonds and 4 single bonds. because of its main component: glass, silicon dioxide is a very common and important molecule in the construction industry. Silicon dioxide, or sio2, has a tetrahedral molecular structure where each silicon atom is bonded to four oxygen atoms. this arrangement forms a strong, stable network found in quartz and glass.
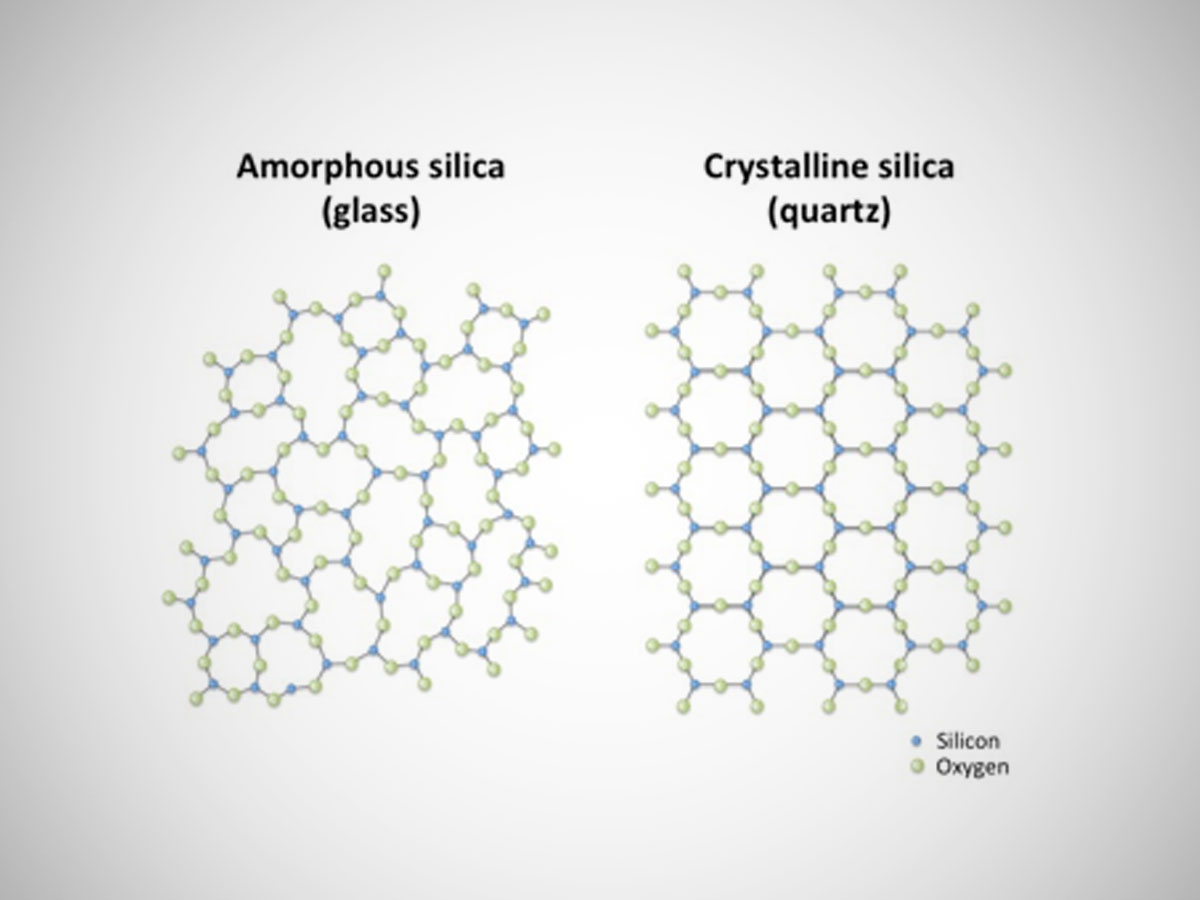
Silicon Dioxide Glass Structure Silicon dioxide, sio a 2, also known as silica is a linear molecule that is formed by one silicon atom and two oxygen atoms with two sets of doubles bonds and 4 single bonds. because of its main component: glass, silicon dioxide is a very common and important molecule in the construction industry. Silicon dioxide, or sio2, has a tetrahedral molecular structure where each silicon atom is bonded to four oxygen atoms. this arrangement forms a strong, stable network found in quartz and glass.

Silicon Dioxide Glass Structure
Comments are closed.