Separating Mixtures Evaporation

Separating Mixtures Evaporation Classx Evaporation is the process of separating a non volatile soluble solid from its mixture in volatile liquid. this process is useful when solid particles do not break down on heating the solution up to the boiling point of the liquid component. In this process, the liquid water is converted into a gas (water vapor) and leaves behind the solid salt. this technique is widely used in various applications, from the production of sea salt to.

Evaporation Separating Mixtures Evaporation Distillation Class 6 Evaporation is the process of the separation of a solid substance that is dissolved in water. the application is based on the fact that solids do not vaporize easily, whereas liquids do. on performing evaporation, the solid substance is left behind as a residue. Evaporation is a technique used to separate out homogeneous mixtures that contain one or more dissolved salts. the method drives off the liquid components from the solid components. the process typically involves heating the mixture until no more liquid remains. Evaporation is a technique for separating mixtures where a liquid is changed into a gas by heating. it can separate solutions by evaporating the solvent like water from salt or sugar solutions. Learn the differences between pure substances and mixtures and how to separate them.
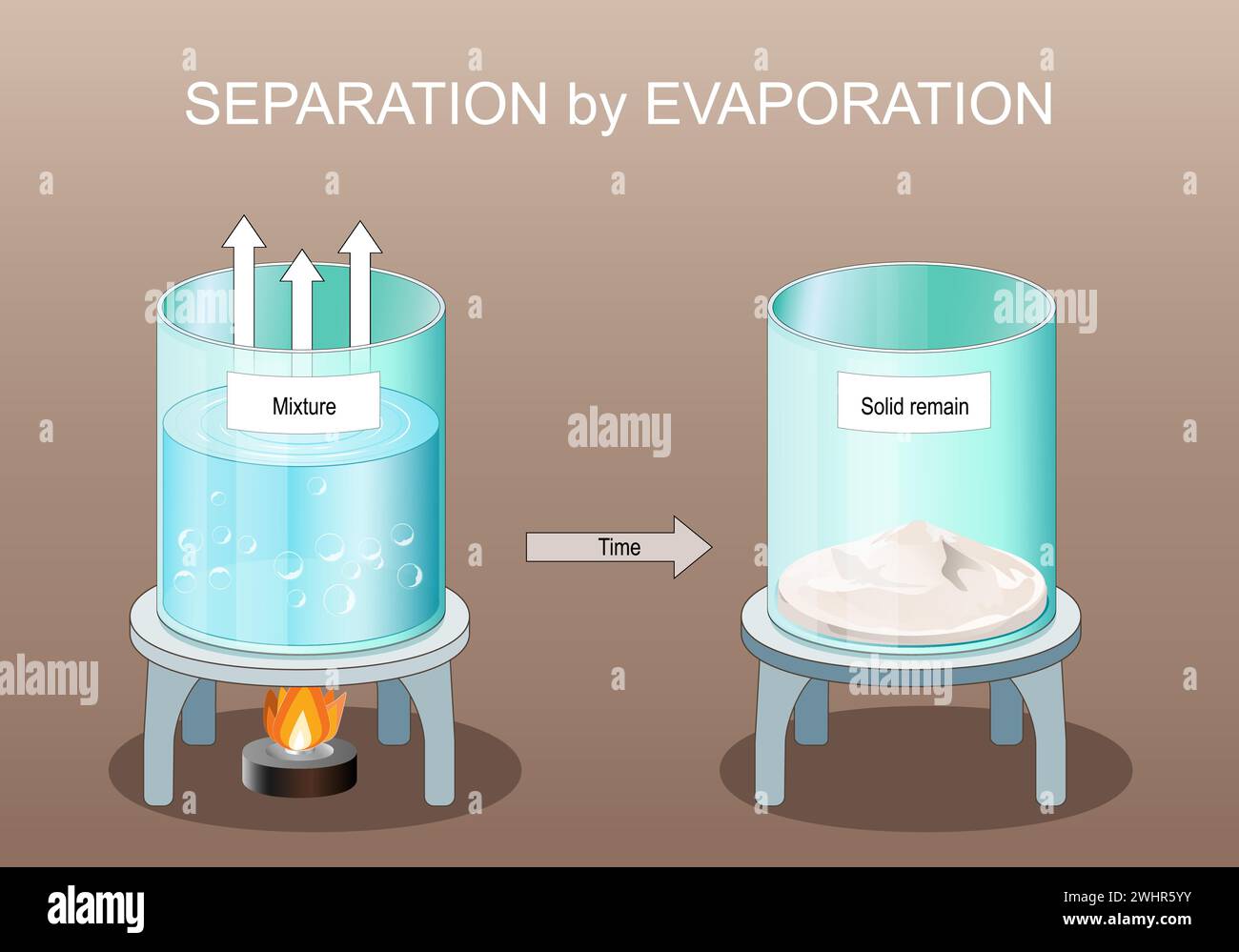
Evaporation Separating Mixtures Evaporation Distillation Class 6 Evaporation is a technique for separating mixtures where a liquid is changed into a gas by heating. it can separate solutions by evaporating the solvent like water from salt or sugar solutions. Learn the differences between pure substances and mixtures and how to separate them. Learn about different methods for separating mixtures, including filtration, distillation, chromatography, evaporation, and crystallisation. understand how each technique works and when to use it. The physical processes of separating mixtures by filtration, evaporation and crystallisation are key to many industrial and research activities. for example, water purification, making edible salts and pharmaceutical formulations all require aspects of these techniques. when research chemists are making new substances, they will often use purification techniques as part of the synthetic. In this lesson, students learn how to separate a mixture of salt and water through the process of evaporation. by creating a saltwater solution and heating it, they observe how the water evaporates, leaving the salt behind, which illustrates the concepts of solute, solvent, and the formation of solutions. Explore filtration, evaporation, and distillation with ngscience. discover practical applications and hands on activities.
Evaporation Separating Mixtures Learn about different methods for separating mixtures, including filtration, distillation, chromatography, evaporation, and crystallisation. understand how each technique works and when to use it. The physical processes of separating mixtures by filtration, evaporation and crystallisation are key to many industrial and research activities. for example, water purification, making edible salts and pharmaceutical formulations all require aspects of these techniques. when research chemists are making new substances, they will often use purification techniques as part of the synthetic. In this lesson, students learn how to separate a mixture of salt and water through the process of evaporation. by creating a saltwater solution and heating it, they observe how the water evaporates, leaving the salt behind, which illustrates the concepts of solute, solvent, and the formation of solutions. Explore filtration, evaporation, and distillation with ngscience. discover practical applications and hands on activities.

Evaporation Separating Mixtures In this lesson, students learn how to separate a mixture of salt and water through the process of evaporation. by creating a saltwater solution and heating it, they observe how the water evaporates, leaving the salt behind, which illustrates the concepts of solute, solvent, and the formation of solutions. Explore filtration, evaporation, and distillation with ngscience. discover practical applications and hands on activities.
Comments are closed.