Selective Precipitation Kf Lecture

Theory Of Size Selective Precipitation Pdf Nanoparticle Solubility We lectured on ch 15 (ksp, ksp and ph and concentration, selective precipitation, kf, complex ion). To understand how several common metal cations can be identified in a solution using selective precipitation.

Modeled Cumulative Precipitation Using Kf2 Solid Lines And Kf1 It discusses the conditions under which pb2 and sr2 precipitate from a solution and the effects of complex ion formation on solubility. additionally, it includes practice problems related to solubility and equilibrium calculations. Watch short videos, explore study materials, and solve practice problems to master key concepts and ace your exams. Example #3: a solution of 0.10 m (each) ni 2 and cu 2 are separated using selective precipitation by the addition of solid na 2 co 3. assuming no volume change upon this addition, how much of the first precipitated ion (in %) remains at the point where the second ion begins to precipitate?. Back to minerals and metal processing: separation of metal cations by selective precipitation • when two metals have salts of the same anion with sufficiently different solubilities, they may be effectively separated by selective precipitation.
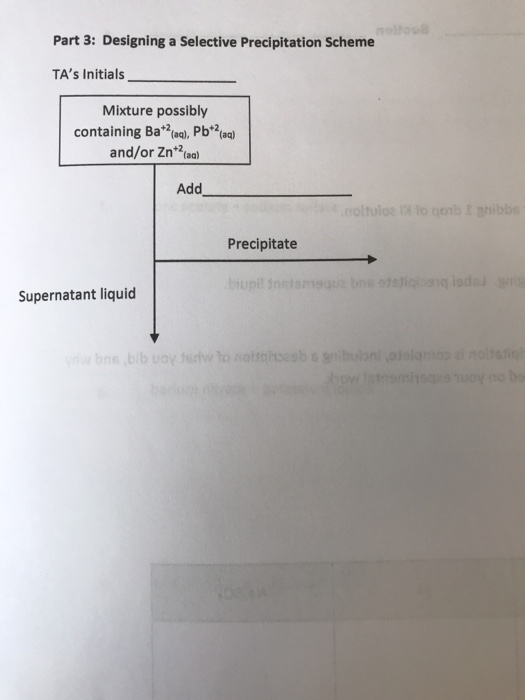
Solved Part 3 Designing A Selective Precipitation Scheme Chegg Example #3: a solution of 0.10 m (each) ni 2 and cu 2 are separated using selective precipitation by the addition of solid na 2 co 3. assuming no volume change upon this addition, how much of the first precipitated ion (in %) remains at the point where the second ion begins to precipitate?. Back to minerals and metal processing: separation of metal cations by selective precipitation • when two metals have salts of the same anion with sufficiently different solubilities, they may be effectively separated by selective precipitation. Thus we can use the ag ions to separate the cl and i ions, a technique called selective precipitation. what would be the [i ] remaining in solution when agcl first precipitates?. Selective precipitation is one of the primary methods for removing these metals before the water can be safely discharged. the two most common approaches are precipitating metals as hydroxides (by raising ph) or as sulfides (by adding sulfur containing compounds). Subscribed 0 45 views 9 months ago we lectured on ch 15 (ksp, selective precipitation, kf, complex ions) more. In solutions containing two or more ions that may form insoluble compounds with the same counter ion, an experimental strategy called selective precipitation may be used to remove individual ions from solution.

Group 1 Precipitation Pdf Thus we can use the ag ions to separate the cl and i ions, a technique called selective precipitation. what would be the [i ] remaining in solution when agcl first precipitates?. Selective precipitation is one of the primary methods for removing these metals before the water can be safely discharged. the two most common approaches are precipitating metals as hydroxides (by raising ph) or as sulfides (by adding sulfur containing compounds). Subscribed 0 45 views 9 months ago we lectured on ch 15 (ksp, selective precipitation, kf, complex ions) more. In solutions containing two or more ions that may form insoluble compounds with the same counter ion, an experimental strategy called selective precipitation may be used to remove individual ions from solution.
Comments are closed.