Science Precigen
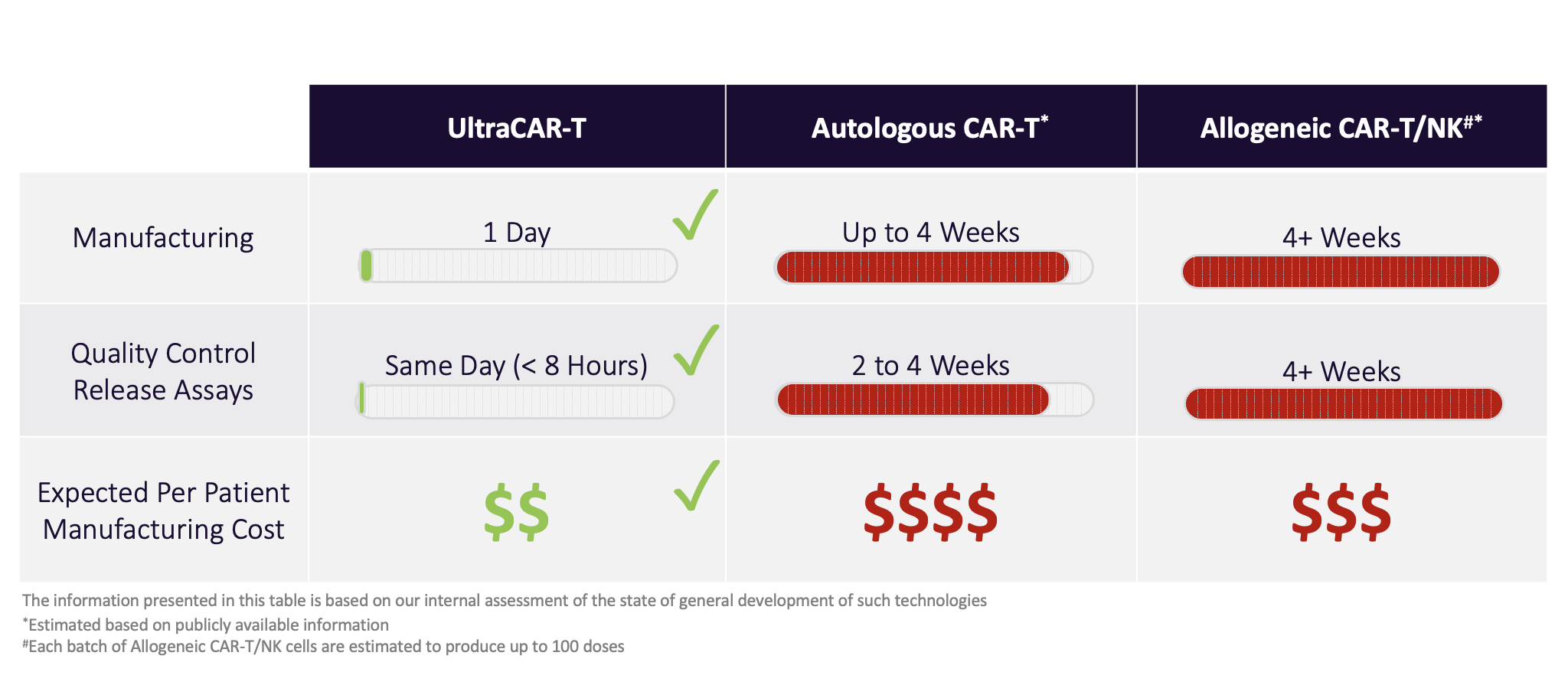
Science Precigen Through innovative science, precigen is redefining what’s possible in precision medicine. the adenoverse platform utilizes a library of proprietary adenovectors for the efficient gene delivery of antigens and cytokines to modulate the immune system. Precigen is a clinical stage biopharmaceutical company based in the united states focused on developing transformative gene and cell therapies that are differentiated not only by their clinical.
Science Precigen Based in germantown, md, precigen is a discovery and clinical stage biotech advancing novel gene and cell therapies which specifically target autoimmune disorders and infectious diseases. Founded with a vision to revolutionize the treatment of complex diseases, precigen has built a robust research and development infrastructure that combines scientific expertise with advanced clinical trial capabilities. Precigen (nasdaq: pgen) is a biopharmaceutical company specializing in the advancement of innovative precision medicines to address difficult to treat diseases with high unmet patient need . Precigen, inc. is a biotech firm dedicated to the research and advancement of synthetic biology technologies. precigen, inc. was established by thomas david reed in 1998 and is headquartered in germantown, maryland.

Science Precigen Precigen (nasdaq: pgen) is a biopharmaceutical company specializing in the advancement of innovative precision medicines to address difficult to treat diseases with high unmet patient need . Precigen, inc. is a biotech firm dedicated to the research and advancement of synthetic biology technologies. precigen, inc. was established by thomas david reed in 1998 and is headquartered in germantown, maryland. Precigen (nasdaq: pgen) is a dedicated discovery and clinical stage biopharmaceutical company advancing the next generation of gene and cell therapies using precision technology to target the. Precigen’s bla was supported by positive data from a pivotal phase i ii trial (nct04724980) that was presented in june at the 2024 american society of clinical oncology (asco) annual meeting, and published last month in the lancet respiratory medicine. The 30 plus year pharma veteran phil tennant is stepping in to take on what could be one of the biggest challenges in life sciences: selling a new gene therapy from a small biotech. Rrp is a rare disease caused by the human papillomavirus (hpv). participants with a pathologically confirmed diagnosis of papilloma and a clinical diagnosis of rrp will be screened for this protocol. this is a nonrandomized, phase 1 2 safety and tolerability study.
Science Precigen Precigen (nasdaq: pgen) is a dedicated discovery and clinical stage biopharmaceutical company advancing the next generation of gene and cell therapies using precision technology to target the. Precigen’s bla was supported by positive data from a pivotal phase i ii trial (nct04724980) that was presented in june at the 2024 american society of clinical oncology (asco) annual meeting, and published last month in the lancet respiratory medicine. The 30 plus year pharma veteran phil tennant is stepping in to take on what could be one of the biggest challenges in life sciences: selling a new gene therapy from a small biotech. Rrp is a rare disease caused by the human papillomavirus (hpv). participants with a pathologically confirmed diagnosis of papilloma and a clinical diagnosis of rrp will be screened for this protocol. this is a nonrandomized, phase 1 2 safety and tolerability study.

Science Precigen The 30 plus year pharma veteran phil tennant is stepping in to take on what could be one of the biggest challenges in life sciences: selling a new gene therapy from a small biotech. Rrp is a rare disease caused by the human papillomavirus (hpv). participants with a pathologically confirmed diagnosis of papilloma and a clinical diagnosis of rrp will be screened for this protocol. this is a nonrandomized, phase 1 2 safety and tolerability study.
Comments are closed.