S Block Elements Oxidesperoxides Superoxide

S Block Elements Pdf Hydroxide Oxide This document discusses the characteristic properties of s block elements, which include the alkali metals (group ia) and alkaline earth metals (group iia). some key points discussed include: s block elements have their outermost shell electrons in the s orbital. Comprehensive jee chemistry guide on s block elements covering electronic configuration, properties, alkali and alkaline earth metals, compounds, trends, and important reactions.

The S Block Elements Pdf Hydroxide Sodium Hydroxide Sodium (na) forms mainly peroxide and some superoxide. potassium (k), rubidium (rb) and cesium (cs) mainly form superoxide because large anions (o 2 ) are stabilized by large cations. Alkaline earth metals react with oxygen to form both normal oxides (e.g., mgo, cao) and peroxides (e.g., sro2, bao2), while alkali metals form oxides, peroxides, and superoxides. For now, we shall study a part of the oxides formed by the elements of the s block in our periodic table and try to understand their nature. that might as well help you out to later innovate new utilities pertaining to these oxides!. The compounds of alkali metals generally reflect the characteristic properties of these elements – their electropositive nature, low ionization enthalpies, and tendency to form 1 ions.
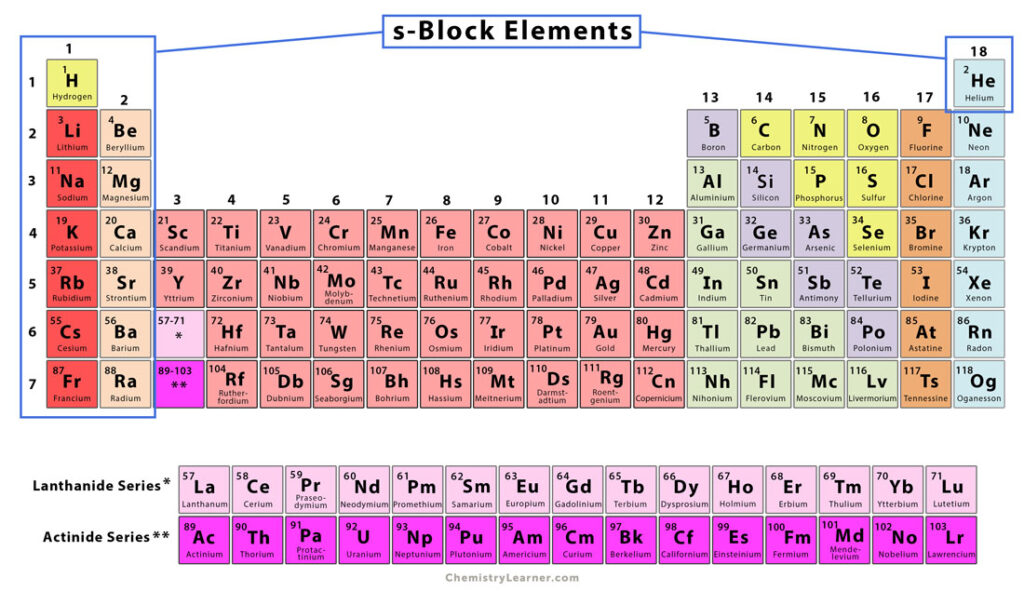
S Block Elements Definition And Characteristics For now, we shall study a part of the oxides formed by the elements of the s block in our periodic table and try to understand their nature. that might as well help you out to later innovate new utilities pertaining to these oxides!. The compounds of alkali metals generally reflect the characteristic properties of these elements – their electropositive nature, low ionization enthalpies, and tendency to form 1 ions. The chemical properties of the s block elements are as fascinating as their physical traits. moreover, these properties are largely influenced by their electronic configuration and position in the periodic table. They have one or two electrons in their outermost s subshell respectively. the elements of group 1 are called alkali metals and those of group 2 are called alkaline earth metals. therefore, alkali metals and alkaline earth metals constitute s block. Elements for which the last electron or valence electrons enter the s subshell are known as s block elements. they are electropositive in nature and lose electrons readily. The s block elements of the periodic table are those in which the last electron enters the outermost s orbital. as the s orbital can accommodate only two electrons, two groups (1 & 2) belong to the s block of the periodic table.

S Block Elements Pdf The chemical properties of the s block elements are as fascinating as their physical traits. moreover, these properties are largely influenced by their electronic configuration and position in the periodic table. They have one or two electrons in their outermost s subshell respectively. the elements of group 1 are called alkali metals and those of group 2 are called alkaline earth metals. therefore, alkali metals and alkaline earth metals constitute s block. Elements for which the last electron or valence electrons enter the s subshell are known as s block elements. they are electropositive in nature and lose electrons readily. The s block elements of the periodic table are those in which the last electron enters the outermost s orbital. as the s orbital can accommodate only two electrons, two groups (1 & 2) belong to the s block of the periodic table.
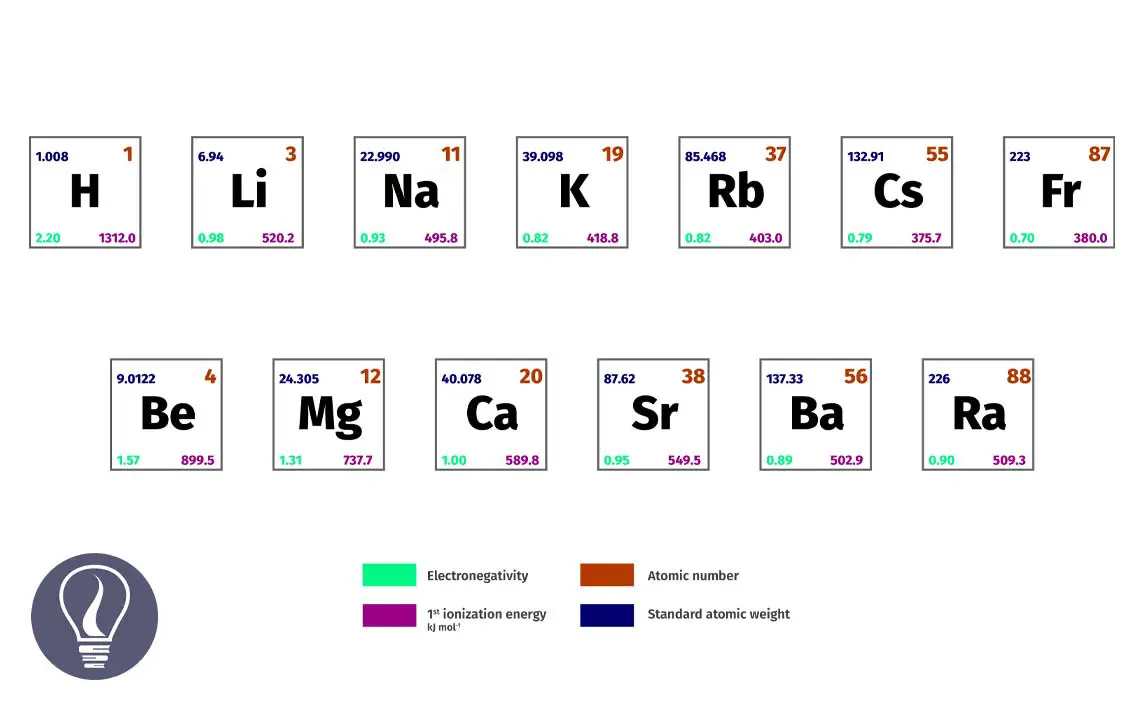
Properties Of S Block Elements Learnbin Elements for which the last electron or valence electrons enter the s subshell are known as s block elements. they are electropositive in nature and lose electrons readily. The s block elements of the periodic table are those in which the last electron enters the outermost s orbital. as the s orbital can accommodate only two electrons, two groups (1 & 2) belong to the s block of the periodic table.

S Block Elements Electronic Configuration And General Properties
Comments are closed.