Reversible Process Vs Irreversible Process In Engineering Key

Irreversible And Reversible Processes Pdf Reversible processes occur without entropy change and can return both system and surroundings to their original states; irreversible processes increase entropy and cannot restore the initial conditions. The distinction between reversible and irreversible processes is fundamental in thermodynamics and mechanical engineering. reversible processes provide an ideal benchmark, while irreversible processes reflect the realities of practical systems, including entropy generation and energy losses.
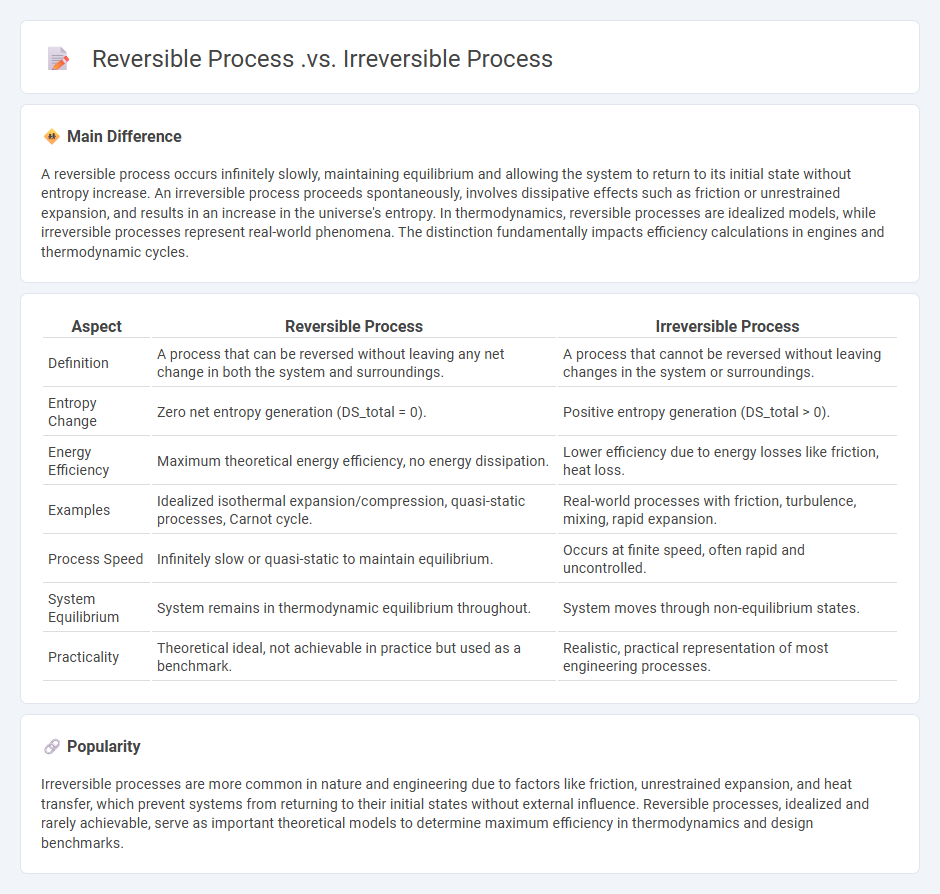
Reversible Process Vs Irreversible Process In Engineering Key The original state of the system and the environment cannot be recreated from the final state in an irreversible process. the coloured states of the system are not in equilibrium with each other on the path of modification from the original state to the final state during the irreversible process. An irreversible process is what we encounter in reality almost all the time. the system and its environment cannot be restored to their original states at the same time. because this is what happens in nature, it is also called a natural process. Irreversible and reversible processes differ in several key attributes, including efficiency, reversibility, entropy change, and practical applications. irreversible processes are characterized by a loss of energy in the form of heat, while reversible processes conserve energy. An irreversible process is what we encounter in reality almost all the time. the system and its environment cannot be restored to their original states at the same time. because this is what happens in nature, it is also called a natural process.

Unit I Reversible Process Irreversibilities Pdf Irreversible and reversible processes differ in several key attributes, including efficiency, reversibility, entropy change, and practical applications. irreversible processes are characterized by a loss of energy in the form of heat, while reversible processes conserve energy. An irreversible process is what we encounter in reality almost all the time. the system and its environment cannot be restored to their original states at the same time. because this is what happens in nature, it is also called a natural process. Reversible and irreversible processes describe how a system changes its state during thermodynamic operations. a reversible process is a perfect, ideal case that can be reversed without any loss. Understanding the difference between reversible and irreversible processes offers valuable insight into the behavior of physical systems, the limits of efficiency, and the natural direction of spontaneous changes in the universe. In these processes a system undergoes through a series of equilibrium states, and when the process is reversed, the system passes through exactly the same equilibrium states. In terms of thermodynamics, a reversible process is where the participants go back to its initial form by inculcating minor or negligible changes in their surroundings. contrarily, an irreversible process is a naturally occurring phenomenon, which does not go back to its original state.

Reversible Process Vs Irreversible Process What S The Difference Reversible and irreversible processes describe how a system changes its state during thermodynamic operations. a reversible process is a perfect, ideal case that can be reversed without any loss. Understanding the difference between reversible and irreversible processes offers valuable insight into the behavior of physical systems, the limits of efficiency, and the natural direction of spontaneous changes in the universe. In these processes a system undergoes through a series of equilibrium states, and when the process is reversed, the system passes through exactly the same equilibrium states. In terms of thermodynamics, a reversible process is where the participants go back to its initial form by inculcating minor or negligible changes in their surroundings. contrarily, an irreversible process is a naturally occurring phenomenon, which does not go back to its original state.

Reversible Process Vs Irreversible Process What S The Difference In these processes a system undergoes through a series of equilibrium states, and when the process is reversed, the system passes through exactly the same equilibrium states. In terms of thermodynamics, a reversible process is where the participants go back to its initial form by inculcating minor or negligible changes in their surroundings. contrarily, an irreversible process is a naturally occurring phenomenon, which does not go back to its original state.
Comments are closed.