Research Development Biologics Development

Research Development Biocon Biologics Because of this reason, we assembled a high level drug product development and manufacturing roadmap. the roadmap is applicable across the different modalities with the intention of providing a unified framework from early phase development to commercialization of biologic drug products. We are grateful to all the partners who commissioned the research, development and manufacturing of their valuable and lifesaving biologics to us in the joint efforts of benefiting global patients.

Gallery Rasayani Biologics Valued at approximately $343 billion in 2021, the global biologics market is expected to reach $569.7 billion by 2027 — a growth of 9.2%.3 this biologics boom is undoubtedly driven by extensive research and development activity. Developing biological products is a complex process that requires multiple steps to launch an effective product. biological products are defined as products containing living microorganisms, such as probiotics or microbial inoculants used in agriculture. these steps include strain discovery to identify active ingredients for a target application, early research and development (r&d) to show. By bridging scientific principles with industrial practices, this comprehensive overview aims to provide a thorough understanding of the various facets of biologics process development, from early stage research to large scale manufacturing. In this article, we explore how an integrated, phase appropriate approach to pharmaceutical development services can enable the successful commercialization of biologics—particularly monoclonal antibodies, therapeutic proteins, antibody drug conjugates, viral vectors, and gene and cell therapies.

Gallery Rasayani Biologics By bridging scientific principles with industrial practices, this comprehensive overview aims to provide a thorough understanding of the various facets of biologics process development, from early stage research to large scale manufacturing. In this article, we explore how an integrated, phase appropriate approach to pharmaceutical development services can enable the successful commercialization of biologics—particularly monoclonal antibodies, therapeutic proteins, antibody drug conjugates, viral vectors, and gene and cell therapies. Most often, the development of a new medicine starts when basic scientists learn of a biological target (e.g., a receptor, enzyme, protein, gene, etc.) that is involved in a biological process thought to be dysfunctional in patients with a disease such as alzheimer's disease (ad). The new center at bridge labs will train the workforce powering north texas’ biotech boom—helping startups speed therapies, vaccines, and breakthrough biologics from lab bench to patients. The roadmap is applicable across the different modalities with the intention of providing a unified framework from early phase development to commercialization of biologic drug products. This section highlights the importance of future directions in biologics development, outlining the emerging trends, the rise of personalized medicine, and the significance of global collaboration in research and development.
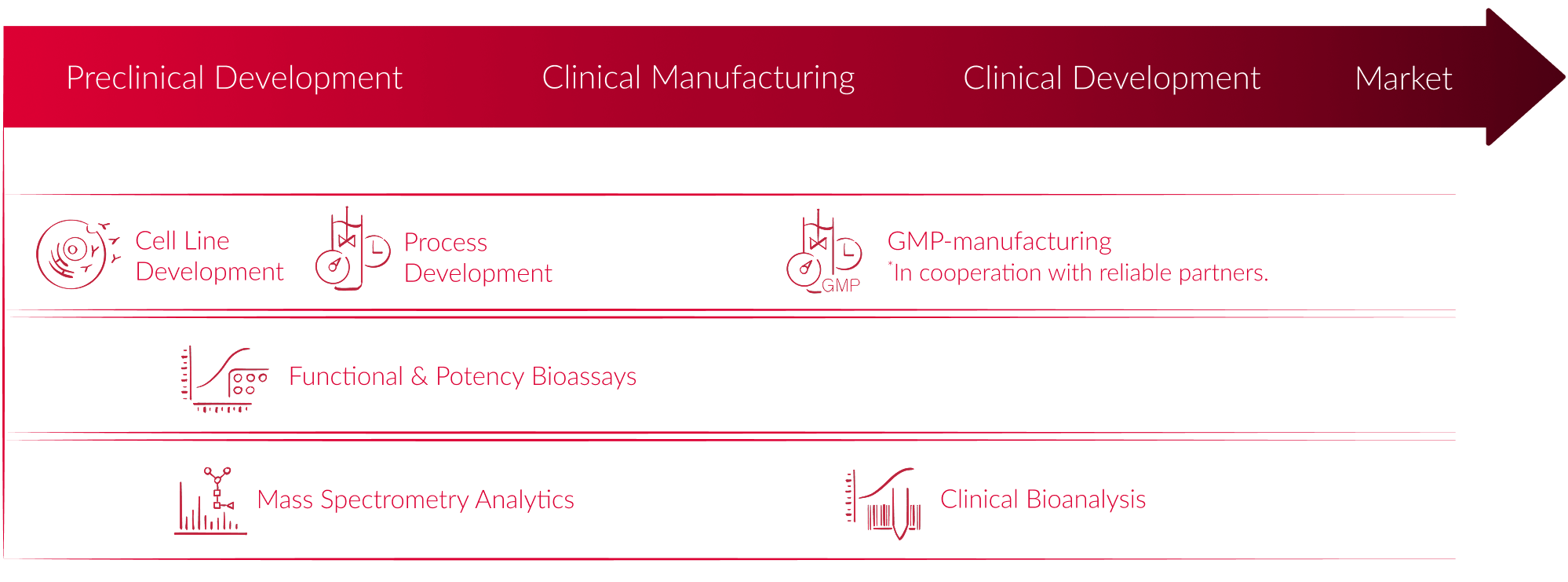
Biologics Development Fyonibio Most often, the development of a new medicine starts when basic scientists learn of a biological target (e.g., a receptor, enzyme, protein, gene, etc.) that is involved in a biological process thought to be dysfunctional in patients with a disease such as alzheimer's disease (ad). The new center at bridge labs will train the workforce powering north texas’ biotech boom—helping startups speed therapies, vaccines, and breakthrough biologics from lab bench to patients. The roadmap is applicable across the different modalities with the intention of providing a unified framework from early phase development to commercialization of biologic drug products. This section highlights the importance of future directions in biologics development, outlining the emerging trends, the rise of personalized medicine, and the significance of global collaboration in research and development.

Advancing Biologics Development With Next Generation Cell Line Development The roadmap is applicable across the different modalities with the intention of providing a unified framework from early phase development to commercialization of biologic drug products. This section highlights the importance of future directions in biologics development, outlining the emerging trends, the rise of personalized medicine, and the significance of global collaboration in research and development.
Comments are closed.