Reducing Sample Size Clinicaltrialist

Reducing Sample To Testing Size Pdf How to reduce sample size for clinical trials one of the questions you will often be asked if you're a clinical trialist is: "can't we make the study smaller?" reducing sample size without losing power can be accomplished by one of three ways. Clinical research validity depends critically on sound sampling methodology and adequate sample size determination, yet many published studies demonstrate deficiencies in these fundamental aspects. this educational review addresses sampling.
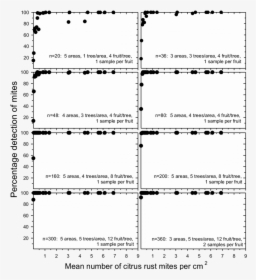
Effect Of Reducing Sample Size On Detecting Citrus Monochrome Hd Png This article reviews basic statistical concepts in sample size estimation, discusses statistical considerations in the choice of a sample size for randomized controlled trials and observational studies, and provides strategies for reducing sample size when planning a study. It is crucial to choose an appropriate sample size estimation to avoid over estimation and under estimation consequences on trial’s outcomes. any negligence in this calculation can result in different types of errors, including approval of false results (type i errors) or rejection of true findings (type ii errors). The statistical adjustments will be demonstrated on case studies in several disease areas as examples of best practices for the use of baseline variables in clinical trial data analysis. How do you reduce sample size in clinical trials? reducing sample size without losing power can be accomplished in one of three principles. this paper examines 15 ways to reduce sample size in clinical trials.

Reducing Sample Size And Obtaining Representative Samples The statistical adjustments will be demonstrated on case studies in several disease areas as examples of best practices for the use of baseline variables in clinical trial data analysis. How do you reduce sample size in clinical trials? reducing sample size without losing power can be accomplished in one of three principles. this paper examines 15 ways to reduce sample size in clinical trials. With the ability to accelerate development timeline, reduce redundant sample size, and select the right dose and patient population during the clinical trial, adaptive designs help to. Sample size re estimation (ssr) is a method used to recalculate sample size during clinical trial conduct to address a lack of adequate information and can have a significant impact on study size, duration, resources, and cost. Reducing sample size without losing power can be accomplished by one of three principles. this paper examines these methods and demonstrates 15 ways to reduce sample size in clinical trials. This article reviews basic statistical concepts in sample size estimation, discusses statistical considerations in the choice of a sample size for randomized controlled trials and observational studies, and provides strategies for reducing sample size when planning a study.
Methods Of Reducing Sample Size In Monte Carlo Computations The With the ability to accelerate development timeline, reduce redundant sample size, and select the right dose and patient population during the clinical trial, adaptive designs help to. Sample size re estimation (ssr) is a method used to recalculate sample size during clinical trial conduct to address a lack of adequate information and can have a significant impact on study size, duration, resources, and cost. Reducing sample size without losing power can be accomplished by one of three principles. this paper examines these methods and demonstrates 15 ways to reduce sample size in clinical trials. This article reviews basic statistical concepts in sample size estimation, discusses statistical considerations in the choice of a sample size for randomized controlled trials and observational studies, and provides strategies for reducing sample size when planning a study.

Effects Of Reducing Sample Size On Model Performance A Cumulative Reducing sample size without losing power can be accomplished by one of three principles. this paper examines these methods and demonstrates 15 ways to reduce sample size in clinical trials. This article reviews basic statistical concepts in sample size estimation, discusses statistical considerations in the choice of a sample size for randomized controlled trials and observational studies, and provides strategies for reducing sample size when planning a study.
Comments are closed.