Quantification Of Related Substances And Unknown Impurities By Hplc

Methods Of Calculations Of Impurities In Hplc Download Free Pdf The presence of related substances in a pharmaceutical product can affect the quality, safety, and efficacy of the product, and it is important to monitor and control their levels. This sop defines the procedure for developing an hplc method to detect and quantify related substances, including known and unknown impurities, degradants, and process related impurities in active pharmaceutical.
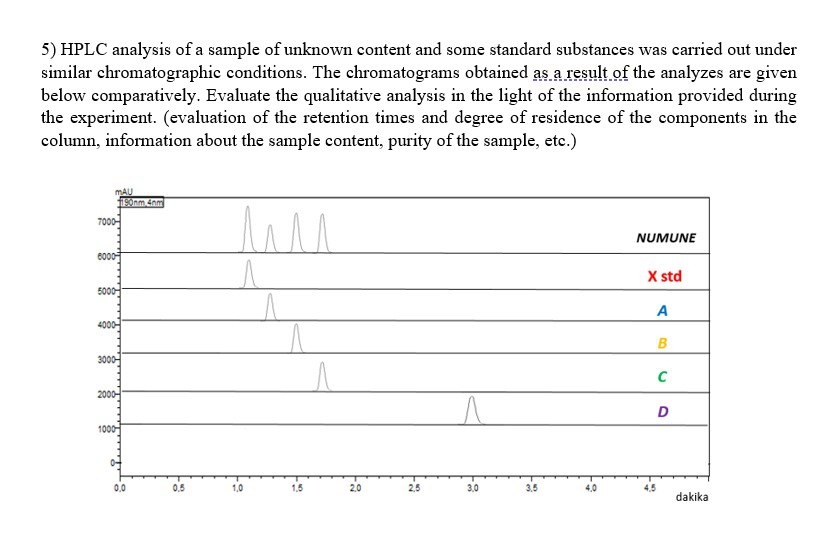
Solved Hplc Analysis Of A Sample Of Unknown Content And Some Chegg A gradient rp hplc method coupled to cad was developed and validated for simultaneous separation and quantification of the nine impurities and other unknown impurities in udca. The approach discussed in this work uses a single hplc platform and provides methods for quantification of known and unknown, nonvolatile residual materials overcoming many of the limitations found with common approaches. The newly developed hplc cad method was successfully applied to commercial bulk drug samples, and two unknown impurities were identified by hplc q tof ms. the effect of cad parameters on the linearity and correction factors was also discussed in this study. A generalized experimental protocol for the analysis of related compounds by hplc is provided below. this protocol should be optimized and validated for each specific api and formulation.

Pdf Validated Gradient Stability Indicating Rp Hplc Method For The The newly developed hplc cad method was successfully applied to commercial bulk drug samples, and two unknown impurities were identified by hplc q tof ms. the effect of cad parameters on the linearity and correction factors was also discussed in this study. A generalized experimental protocol for the analysis of related compounds by hplc is provided below. this protocol should be optimized and validated for each specific api and formulation. This comprehensive review provides an in depth, methodical exploration of the analytical method development process specifically tailored for these impurities, with a pronounced emphasis on high performance liquid chromatography (hplc) due to its unparalleled versatility and quantitative precision. Acceptance criteria. the obtained results are reported, highlighting the percentage of related substances and any observed impurities or degradants. During the hands on session, participants will perform testing with hplc for organic impurities and related substances in dosage forms or drug substances. participants will also manually calculate the results after performing manual integration of the hplc measurements. In the present study, a stability indicating, selective, sensitive validating approach with a high performance liquid chromatography (hplc) technique is used to determine the impurities specification limit at a very subtle level of below 0.1% with a short run time in the elvitegravir drug substance.

Pdf A Validated Rp Hplc Method For Quantitative Determination Of This comprehensive review provides an in depth, methodical exploration of the analytical method development process specifically tailored for these impurities, with a pronounced emphasis on high performance liquid chromatography (hplc) due to its unparalleled versatility and quantitative precision. Acceptance criteria. the obtained results are reported, highlighting the percentage of related substances and any observed impurities or degradants. During the hands on session, participants will perform testing with hplc for organic impurities and related substances in dosage forms or drug substances. participants will also manually calculate the results after performing manual integration of the hplc measurements. In the present study, a stability indicating, selective, sensitive validating approach with a high performance liquid chromatography (hplc) technique is used to determine the impurities specification limit at a very subtle level of below 0.1% with a short run time in the elvitegravir drug substance.
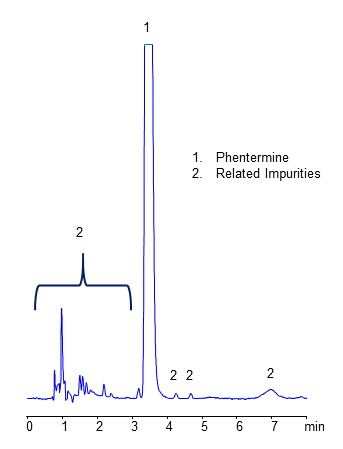
Hplc Methods For Analysis Of Related Impurities Helix Chromatography During the hands on session, participants will perform testing with hplc for organic impurities and related substances in dosage forms or drug substances. participants will also manually calculate the results after performing manual integration of the hplc measurements. In the present study, a stability indicating, selective, sensitive validating approach with a high performance liquid chromatography (hplc) technique is used to determine the impurities specification limit at a very subtle level of below 0.1% with a short run time in the elvitegravir drug substance.
Comments are closed.