Q A How Cepi Funded Research Is Supporting The Covid 19 Vaccine
Q A How Cepi Funded Research Is Supporting The Covid 19 Vaccine Following two scientific calls to find clinical trial partners in january and october 2021, cepi has pushed forward a series of studies, supported by up to $80m in funding, to provide scientists and decision makers with more in depth knowledge on how covid 19 vaccines work. Following two scientific calls to find clinical trial partners in january and october 2021, cepi has pushed forward a series of studies, supported by up to $80m in funding, to provide scientists and decision makers with more in depth knowledge on how covid 19 vaccines work.

Q A How Cepi Funded Research Is Supporting The Covid 19 Vaccine This article highlights and evaluates the role of cepi and its contribution to global equitable access to covid 19 vaccines through its established partnerships for vaccine development. In the months that followed, cepi built one of the world’s largest and most diverse covid 19 vaccine portfolios, based on the principles of speed, scale, and access. cepi invested over us $1.5 billion to advance 14 covid 19 vaccines, seven of which have been approved for domestic or global use. Over the past 2 years, cepi has spent almost $2 billion in mostly public funding to support more than a dozen promising covid 19 vaccine candidates. the idea was that cepi funded manufacturers of successful vaccines would supply doses through covax, thereby ensuring access for all. Over the past 13 months, glopid r observer cepi has been working at the forefront of covid 19 vaccine r&d, committing over us $1 billion into 11 diverse partnerships to build the world’s largest covid 19 vaccine portfolio.
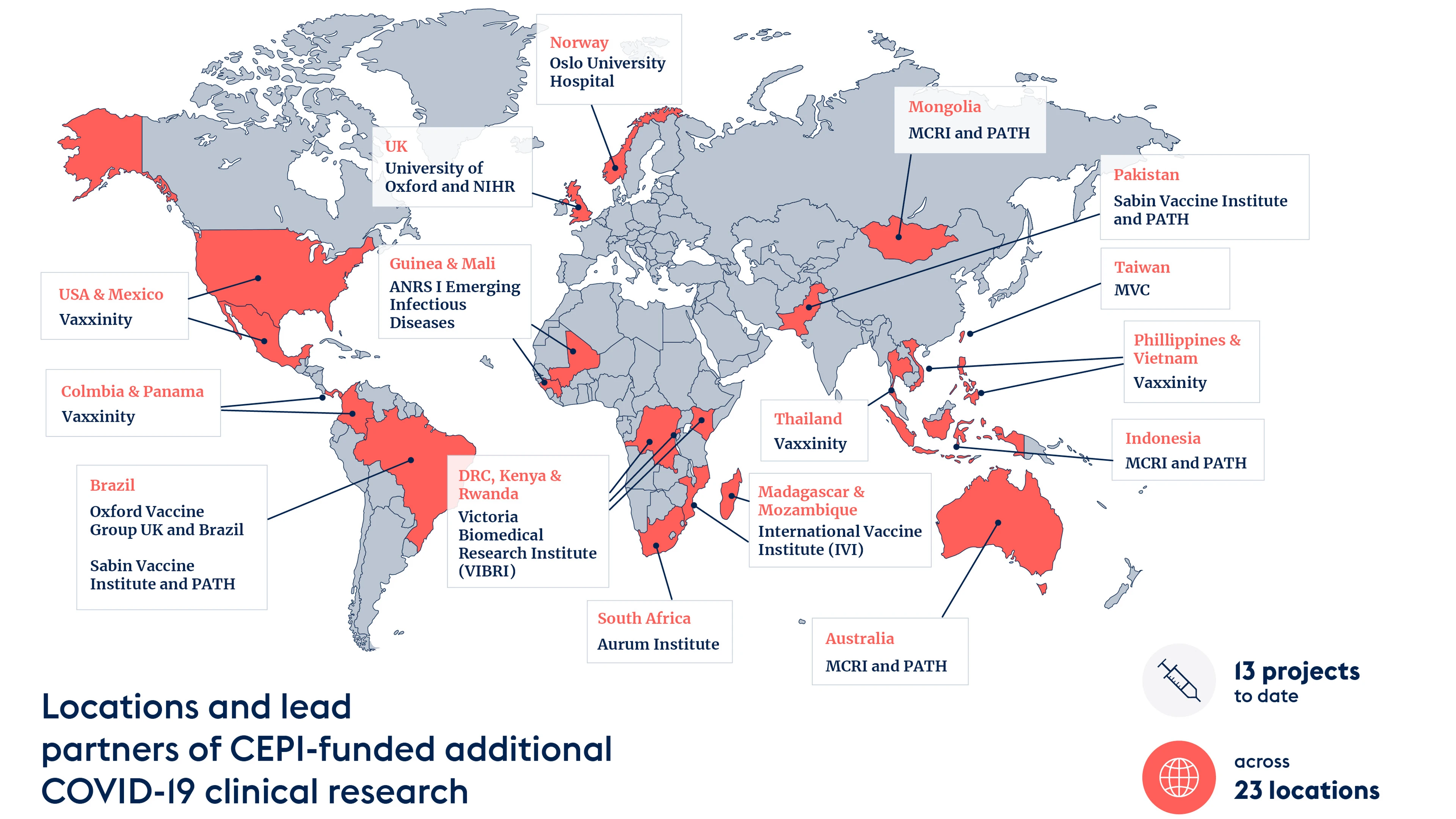
Q A How Cepi Funded Research Is Supporting The Covid 19 Vaccine Over the past 2 years, cepi has spent almost $2 billion in mostly public funding to support more than a dozen promising covid 19 vaccine candidates. the idea was that cepi funded manufacturers of successful vaccines would supply doses through covax, thereby ensuring access for all. Over the past 13 months, glopid r observer cepi has been working at the forefront of covid 19 vaccine r&d, committing over us $1 billion into 11 diverse partnerships to build the world’s largest covid 19 vaccine portfolio. The cepi centralised laboratory network aims to enable key immunogenicity and efficacy endpoint evaluation, support covid 19 vaccine developers in the pathway towards licensure, and help the identification of immune correlates of protection. Within approximately 2 years, the cepi cln provided preclinical to phase 3 clinical trial support to more than 50 vaccine developers (some funded by the covax initiative) and allocated more. The cepi centralized laboratory network (cln) has significantly contributed to the development of several approved sars cov 2 vaccines by conducting over 70,000 clinical samples for testing from various vaccine developers. In a new q a blog post, cepi’s director of clinical development, jakob cramer, outlines the studies we are supporting to better inform global covid 19 vaccination strategies and what.

Canada Boosts Funding For Cepi S Covid 19 Vaccine Research The cepi centralised laboratory network aims to enable key immunogenicity and efficacy endpoint evaluation, support covid 19 vaccine developers in the pathway towards licensure, and help the identification of immune correlates of protection. Within approximately 2 years, the cepi cln provided preclinical to phase 3 clinical trial support to more than 50 vaccine developers (some funded by the covax initiative) and allocated more. The cepi centralized laboratory network (cln) has significantly contributed to the development of several approved sars cov 2 vaccines by conducting over 70,000 clinical samples for testing from various vaccine developers. In a new q a blog post, cepi’s director of clinical development, jakob cramer, outlines the studies we are supporting to better inform global covid 19 vaccination strategies and what.
Cepi Funded Covid 19 Vaccine Candidates Progress To Clinical Trials Cepi The cepi centralized laboratory network (cln) has significantly contributed to the development of several approved sars cov 2 vaccines by conducting over 70,000 clinical samples for testing from various vaccine developers. In a new q a blog post, cepi’s director of clinical development, jakob cramer, outlines the studies we are supporting to better inform global covid 19 vaccination strategies and what.
Comments are closed.