Precigen Pgen Is Up 96 8 After Fda Fully Approves Papzimeos As First
Precigen Pgen Is Up 96 8 After Fda Fully Approves Papzimeos As First With the landmark fda approval of papzimeos and broad label, all adult rrp patients are now eligible for access to the first and only approved therapy that targets the root cause of the disease," said helen sabzevari, phd, president and ceo of precigen. Precigen completed submission of the rolling biologics license application (bla) in december 2024 under an accelerated approval pathway; however, the fda has granted papzimeos full.
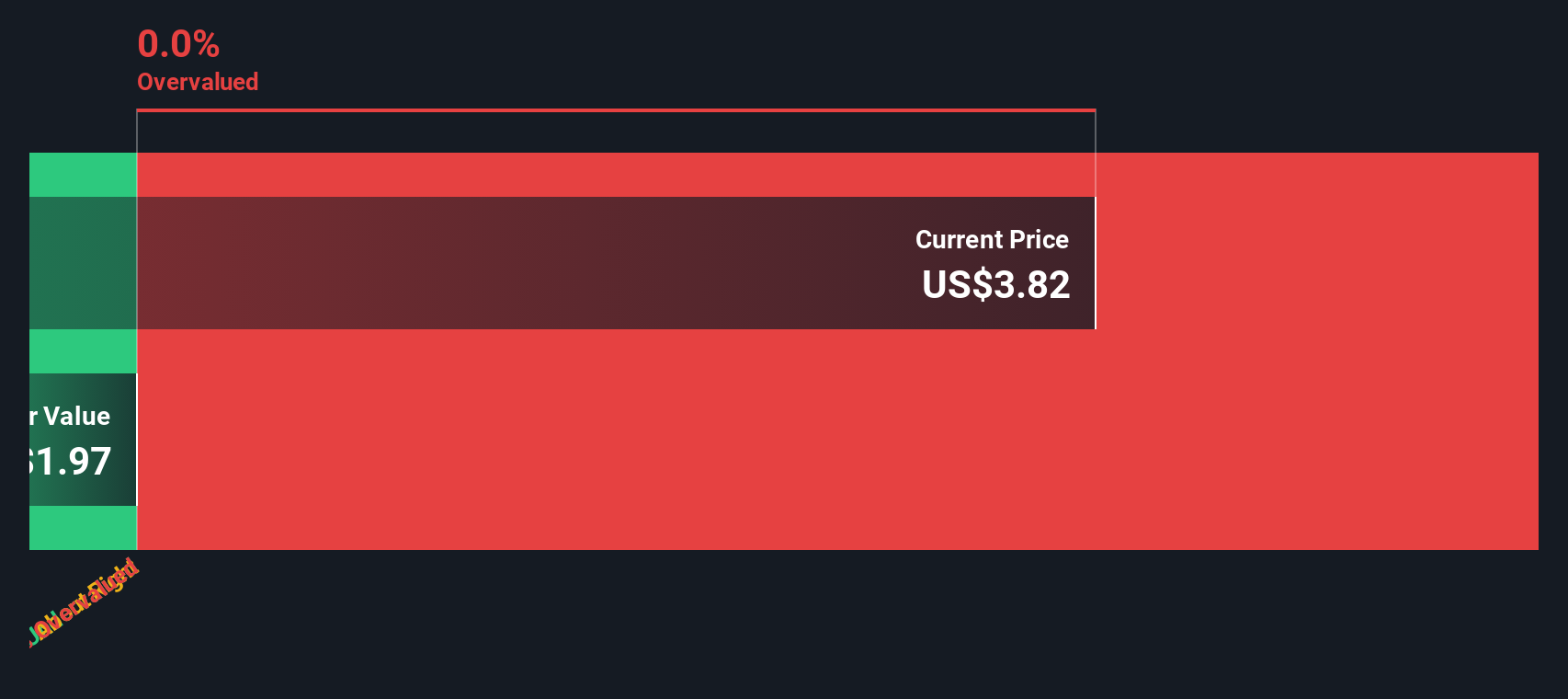
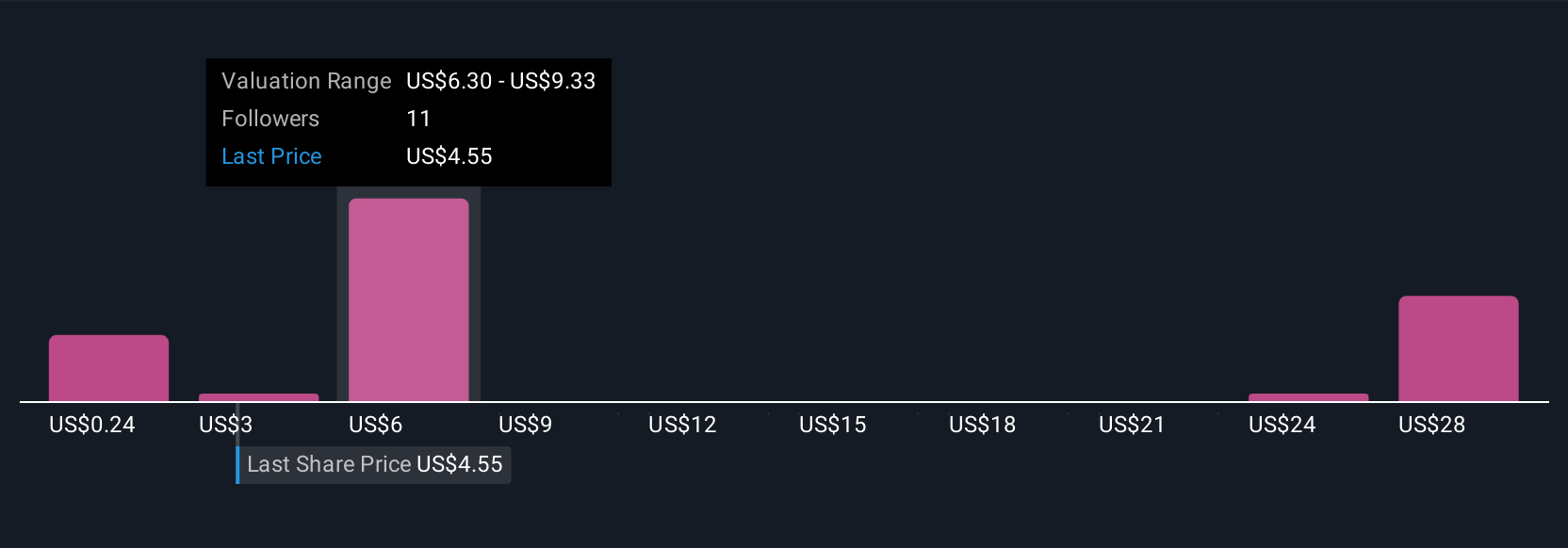
Precigen Pgen Valuation Insights After Fda Greenlights First In The approval is based on results from a single arm, open label trial evaluating papzimeos in adult patients with rrp who required three or more surgeries per year. With the landmark fda approval of papzimeos and broad label, all adult rrp patients are now eligible for access to the first and only approved therapy that targets the root cause of the. In august 2025, precigen achieved a historic milestone with the fda’s full approval of papzimeos™ (zopapogene imadenovec drba), the first and only therapy approved for adults suffering from recurrent respiratory papillomatosis (rrp). Key takeaway: precigen, inc. announced full fda approval for papzimeos (zopapogene imadenovec drba), the first and only therapy for adults with recurrent respiratory papillomatosis (rrp).

Precigen Pgen Valuation Insights After Fda Greenlights First In In august 2025, precigen achieved a historic milestone with the fda’s full approval of papzimeos™ (zopapogene imadenovec drba), the first and only therapy approved for adults suffering from recurrent respiratory papillomatosis (rrp). Key takeaway: precigen, inc. announced full fda approval for papzimeos (zopapogene imadenovec drba), the first and only therapy for adults with recurrent respiratory papillomatosis (rrp). The approval of the first non surgical treatment for adults with rrp marks not only a scientific milestone, but an inflection point for the entire rrp community. Precigen announces full fda approval of papzimeos (zopapogene imadenovec drba), the first and only approved therapy for the treatment of adults with recurrent respiratory papillomatosis. And although papzimeos was once on a path to accelerated approval, which would require a post market confirmatory trial, the fda instead gave it a full nod. what’s more, the august decision came almost two weeks earlier than the scheduled pdufa date. “this is somewhat unprecedented,” sabzevari said. Precigen's immunotherapy for a rare respiratory disease has become the first treatment to win u.s. regulatory approval for the condition, which typically requires frequent surgeries,.
Comments are closed.