Precigen Fireside Chat Papzimeos Launch And First Line Rrp Standard Of Care Update March 31 2026

Precigen Secures Fda Approval For Papzimeos And Redefines Rrp Treatment Precigen (nasdaq:pgen) executives used a fireside chat hosted by h.c. wainwright to outline the company’s transition into a commercial stage organization following the fda approval of papzimeos, the first immunotherapy approved for adults with recurrent respiratory papillomatosis (rrp). So as we sit here in late march of 2026, the company has officially transitioned into a commercial stage entity reporting its first quarter of launch very recently.
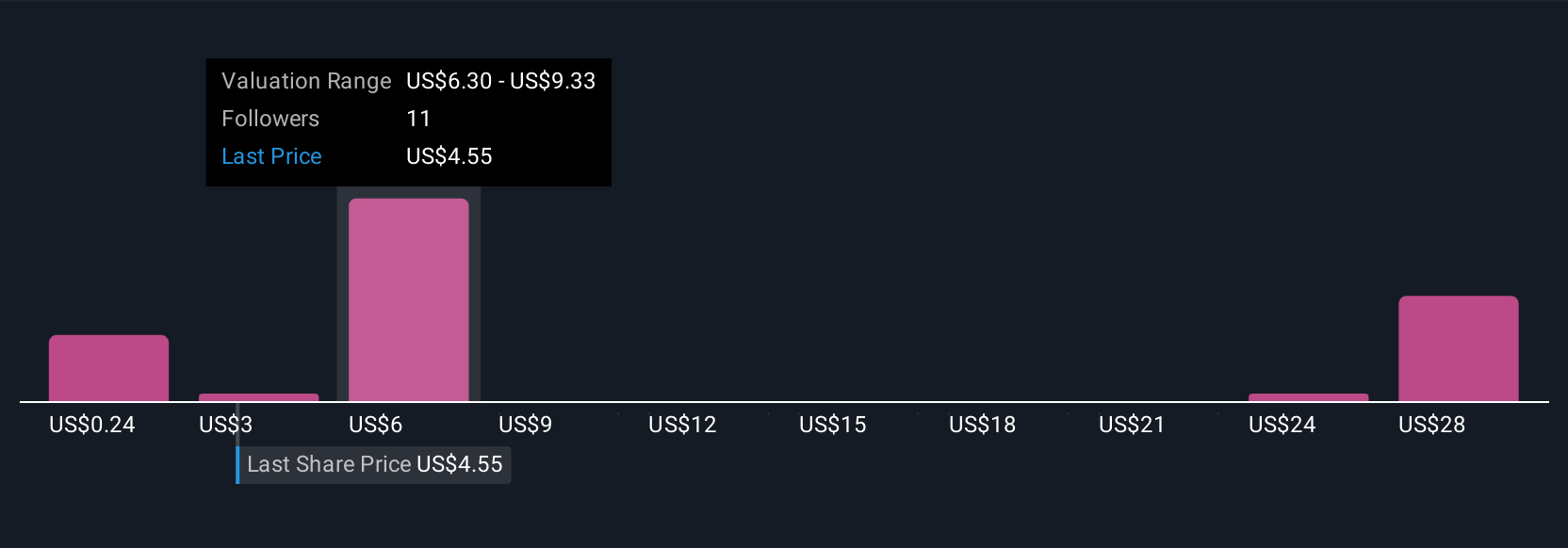
Precigen Pgen Is Up 96 8 After Fda Fully Approves Papzimeos As First Precigen fireside chat | papzimeos launch and first line rrp standard of care update.twitter twitter i101inif you find our work useful, please. Precigen (nasdaq:pgen) executives used a fireside chat hosted by h.c. wainwright to outline the company’s transition into a commercial stage organization following the fda approval of. Experts view papzimeos as a blockbuster success, solidifying its position as the first line standard of care for adults with rrp, while analysts anticipate sustained revenue growth and long term viability for precigen. A significant january 2026 consensus paper, authored by 16 leading rrp physicians, officially recommended papzimeos as the first line standard for adults with rrp.
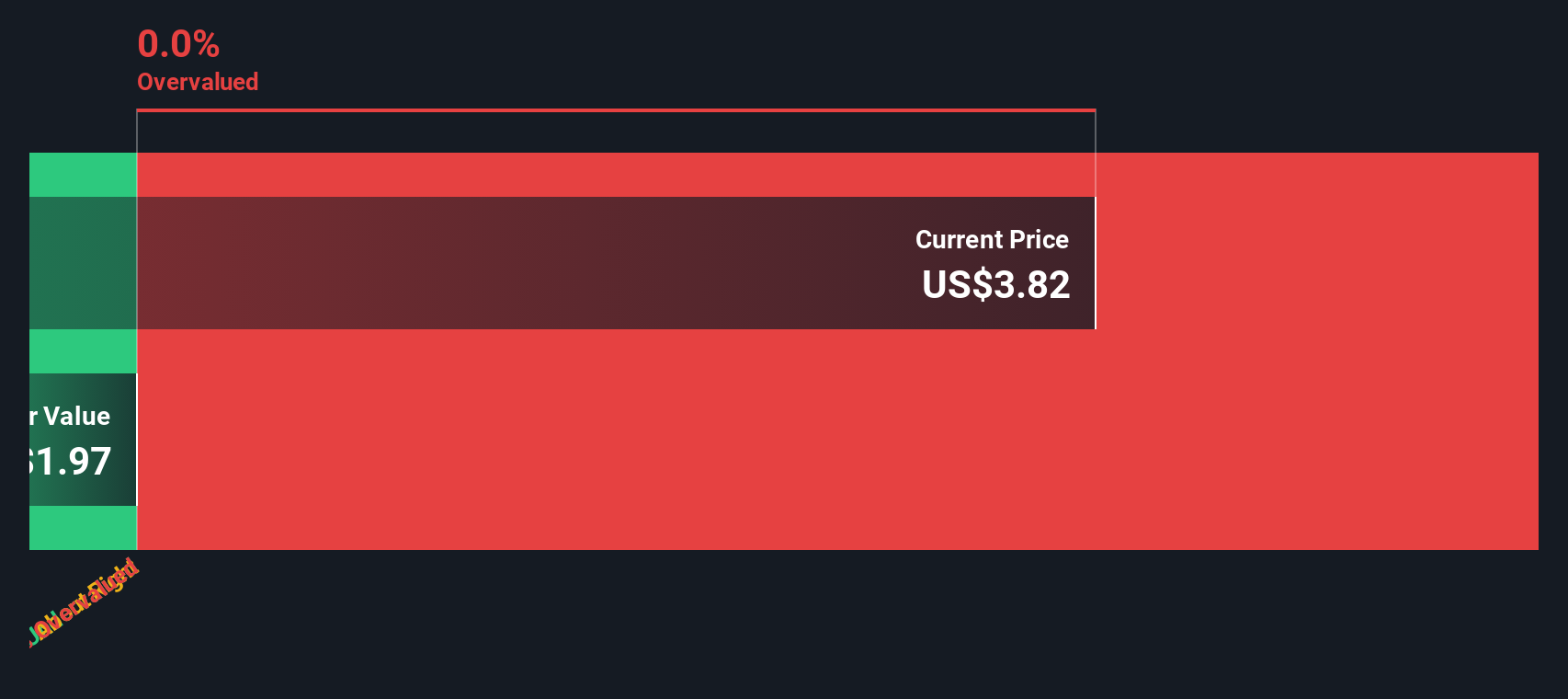
Precigen Pgen Valuation Insights After Fda Greenlights First In Experts view papzimeos as a blockbuster success, solidifying its position as the first line standard of care for adults with rrp, while analysts anticipate sustained revenue growth and long term viability for precigen. A significant january 2026 consensus paper, authored by 16 leading rrp physicians, officially recommended papzimeos as the first line standard for adults with rrp. New expert consensus published in the laryngoscope recommends papzimeos (zopapogene imadenovec) as the new standard of care first line treatment for adults with recurrent respiratory papillomatosis. "we are seeing strong alignment within the physician community around papzimeos as the first line standard of care for adults with rrp, supported by its profile as the only approved. As a result of its transformative clinical data, papzimeos has been endorsed by expert consensus papers sponsored by the rrp foundation, recommending it as the first immunotherapy and preferred first line treatment for adults with recurrent respiratory papillomatosis. Precigen to participate in a fireside chat with h.c. wainwright on march 31 prnewswire • 03 30 26.

Precigen Pgen Valuation Insights After Fda Greenlights First In New expert consensus published in the laryngoscope recommends papzimeos (zopapogene imadenovec) as the new standard of care first line treatment for adults with recurrent respiratory papillomatosis. "we are seeing strong alignment within the physician community around papzimeos as the first line standard of care for adults with rrp, supported by its profile as the only approved. As a result of its transformative clinical data, papzimeos has been endorsed by expert consensus papers sponsored by the rrp foundation, recommending it as the first immunotherapy and preferred first line treatment for adults with recurrent respiratory papillomatosis. Precigen to participate in a fireside chat with h.c. wainwright on march 31 prnewswire • 03 30 26.

Precigen S Fda Approval Of Papzimeos A Game Changer For The Rrp Market As a result of its transformative clinical data, papzimeos has been endorsed by expert consensus papers sponsored by the rrp foundation, recommending it as the first immunotherapy and preferred first line treatment for adults with recurrent respiratory papillomatosis. Precigen to participate in a fireside chat with h.c. wainwright on march 31 prnewswire • 03 30 26.
Comments are closed.