Physical Science Atoms Elements

Science 9 Physical Science Atoms And Elements Atoms The document explores the evolution of atomic and elemental concepts from ancient greece to the present, detailing contributions by prominent scientists and alchemists. What is an atom? an atom is the basic building block of chemistry. it is the smallest unit into which matter can be divided without the release of electrically charged particles. it also is the smallest unit of matter that has the characteristic properties of a chemical element.

Atoms And Elements Physical Science Ppt Pptx All matter in the natural world is composed of one or more of the 92 fundamental substances called elements. an element is a pure substance that is distinguished from all other matter by the fact that it cannot be created or broken down by ordinary chemical means. Atoms consist of a dense nucleus made of protons and neutrons, surrounded by electrons. isotopes are atoms of the same element that have different numbers of neutrons; some isotopes are stable, while others are radioactive. An atom is the smallest unit of an element that retains its chemical properties. it is made of protons, neutrons, and electrons. (image: m. magnaye) atoms are the building blocks of matter. everything around us — from air and water, to rocks, plants and animals — as well as everything within our bodies, is made up of atoms. Matter is composed of exceedingly small particles called atoms. an atom is the smallest unit of an element that can participate in a chemical change. an element consists of only one type of atom, which has a mass that is characteristic of the element and is the same for all atoms of that element.

Science 90 Physical Science Atoms And Elements Chemistry An atom is the smallest unit of an element that retains its chemical properties. it is made of protons, neutrons, and electrons. (image: m. magnaye) atoms are the building blocks of matter. everything around us — from air and water, to rocks, plants and animals — as well as everything within our bodies, is made up of atoms. Matter is composed of exceedingly small particles called atoms. an atom is the smallest unit of an element that can participate in a chemical change. an element consists of only one type of atom, which has a mass that is characteristic of the element and is the same for all atoms of that element. This section introduces the basic building blocks of matter: atoms and elements, how they are organized in the periodic table, and how chemists use the mole to count extremely large numbers of particles. Atoms are the building blocks of matter. they are the smallest particles of an element that still have the element's properties. elements, in turn, are pure substances—such as nickel, hydrogen, and helium—that make up all kinds of matter. Atoms consist of three basic particles: protons, electrons, and neutrons. the nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). the outermost regions of the atom are called electron shells and contain the electrons (negatively charged). All matter is made up of substances called elements, which have specific chemical and physical properties and cannot be broken down into other substances through ordinary chemical reactions. gold, for instance, is an element, and so is carbon. there are 118 elements, but only 92 occur naturally.
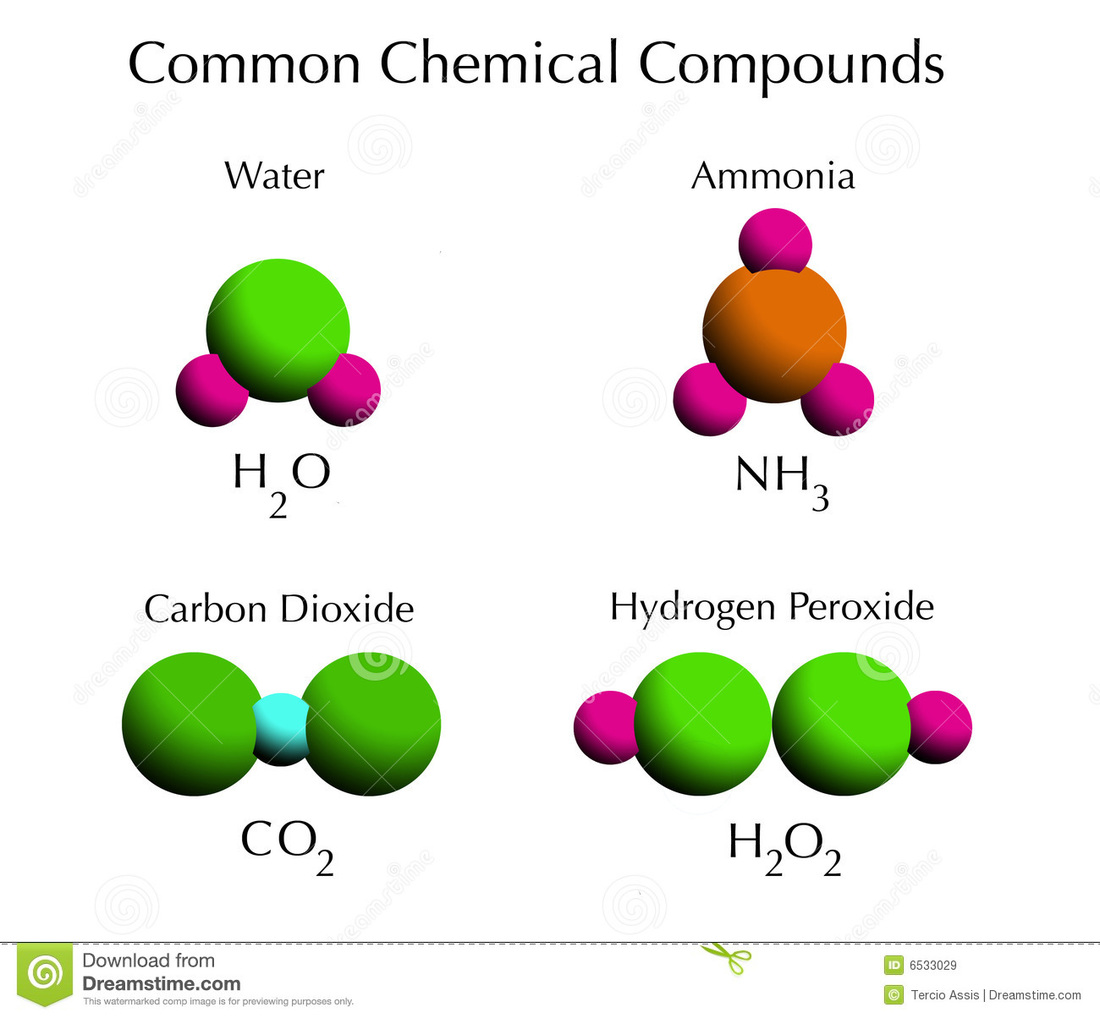
Atoms Elements Compounds This section introduces the basic building blocks of matter: atoms and elements, how they are organized in the periodic table, and how chemists use the mole to count extremely large numbers of particles. Atoms are the building blocks of matter. they are the smallest particles of an element that still have the element's properties. elements, in turn, are pure substances—such as nickel, hydrogen, and helium—that make up all kinds of matter. Atoms consist of three basic particles: protons, electrons, and neutrons. the nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). the outermost regions of the atom are called electron shells and contain the electrons (negatively charged). All matter is made up of substances called elements, which have specific chemical and physical properties and cannot be broken down into other substances through ordinary chemical reactions. gold, for instance, is an element, and so is carbon. there are 118 elements, but only 92 occur naturally.

Grade 7 Science Physical Science Atoms Elements Periodic Table Ngss Atoms consist of three basic particles: protons, electrons, and neutrons. the nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). the outermost regions of the atom are called electron shells and contain the electrons (negatively charged). All matter is made up of substances called elements, which have specific chemical and physical properties and cannot be broken down into other substances through ordinary chemical reactions. gold, for instance, is an element, and so is carbon. there are 118 elements, but only 92 occur naturally.
Comments are closed.