Phase I Ii Study Of Lyl314 A Novel Cd19 Cd20 Targeting Car T Cell For R R Lbcl
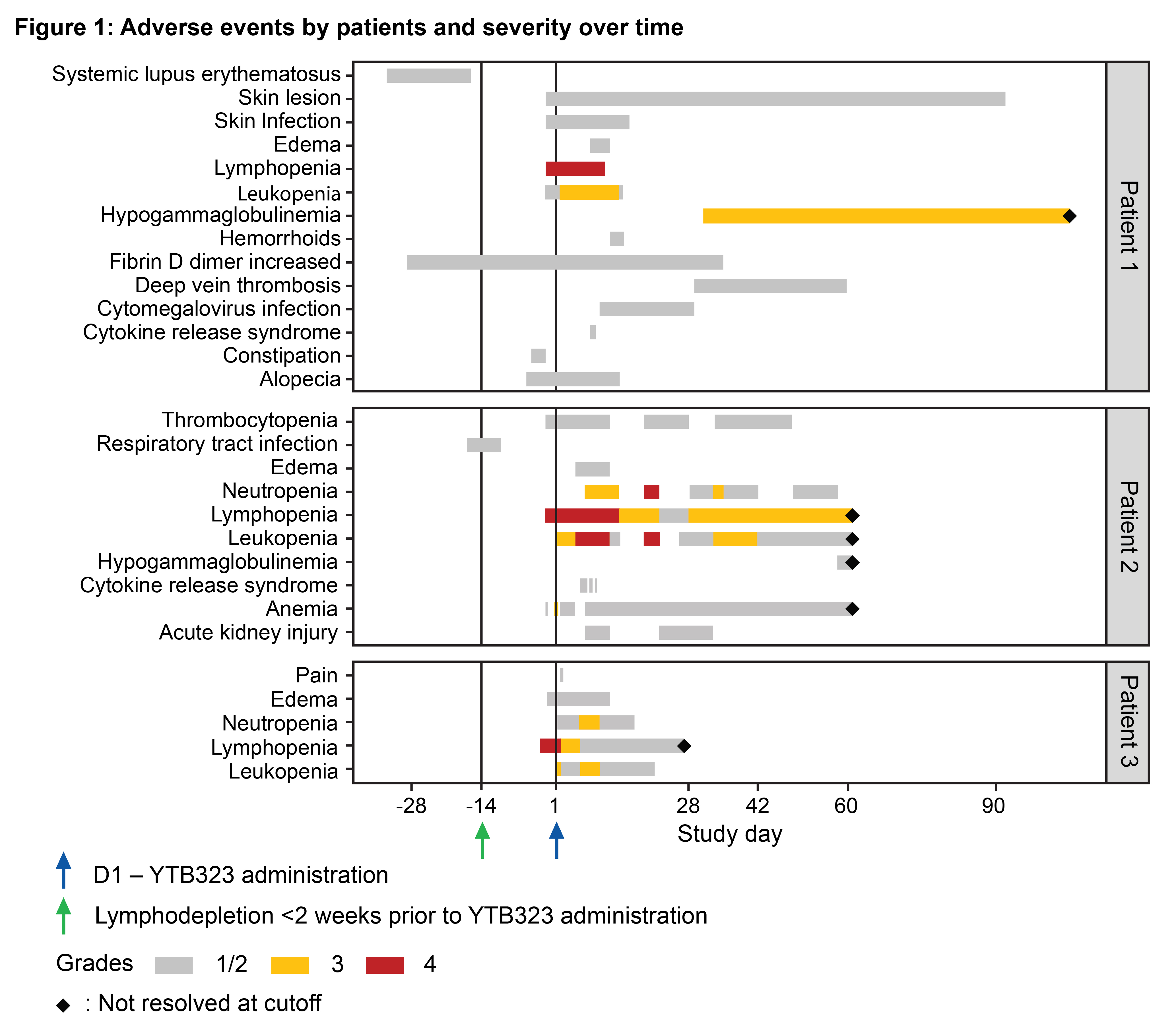
An Open Label Multicenter Phase 1 2 Study To Assess Safety Efficacy Lyl314 (formerly impt 314) is a next generation dual targeting cd19 cd20 car t cell product candidate designed to increase complete response rates and prolong the duration of the responses as compared to the approved cd19‑targeted car t cell therapies for the treatment of large b cell lymphoma. Conclusions: a high overall response rate (94%) and complete response rate (74%) with a manageable safety profile were achieved with lyl314 in patients with r r lbcl in an ongoing phase 1 2 multi center trial.

Bispecific Anti Cd20 Anti Cd19 Car T Cells For Relapsed B Cell This is a phase 1 2, multi center, open label study evaluating the safety and efficacy of rondecabtagene autoleucel (ronde cel) also known as lyl314, a dual targeting chimeric antigen receptor (car) targeting cluster of differentiation (cd)19 and cd20 in participants with aggressive large b cell lymphoma. Lyl314 (formerly impt 314) is a next generation dual targeting cd19 cd20 car t cell product candidate designed to increase complete response rates and prolong the duration of the responses as compared to the approved cd19‑targeted car t cell therapies for the treatment of large b cell lymphoma. This is a phase 1 2, multi center, open label study evaluating the safety and efficacy of lyl314, a dual targeting chimeric antigen receptor (car) targeting cluster of differentiation (cd)19 and cd20 in participants with aggressive large b cell lymphoma. Akil merchant, md, cedars sinai medical center, los angeles, ca, shares insights into a phase i ii trial exploring lyl314, a cd19 cd20 targeting car t cell, for patients with relapsed refractory (r r) large b cell lymphoma (lbcl).

Pdf Secondary Myeloid Neoplasms After Cd19 Car T Therapy In Patients This is a phase 1 2, multi center, open label study evaluating the safety and efficacy of lyl314, a dual targeting chimeric antigen receptor (car) targeting cluster of differentiation (cd)19 and cd20 in participants with aggressive large b cell lymphoma. Akil merchant, md, cedars sinai medical center, los angeles, ca, shares insights into a phase i ii trial exploring lyl314, a cd19 cd20 targeting car t cell, for patients with relapsed refractory (r r) large b cell lymphoma (lbcl). Lyell immunopharma announced positive new clinical data demonstrating high rates of durable complete responses from the phase 1 2 trial of its dual targeting cd19 cd20 car t cell candidate, including data from patients with large b cell lymphoma (lbcl) treated in the third or later line setting. Lyl314 (formerly impt 314) is a next generation dual targeting cd19 cd20 car t cell product candidate designed to increase complete response rates and prolong the duration of the responses as compared to the approved cd19‑targeted car t cell therapies for the treatment of large b cell lymphoma. Lyell immunopharma has reported new clinical data from the phase i ii clinical trial of autologous dual targeting cd19 cd20 chimeric antigen receptor (car) t cell product candidate, lyl314, along with data from those with large b cell lymphoma (lbcl) in the third or later line (3l ) setting.
Comments are closed.