Phase Diagram Thermodynamics At Darcy Bromley Blog
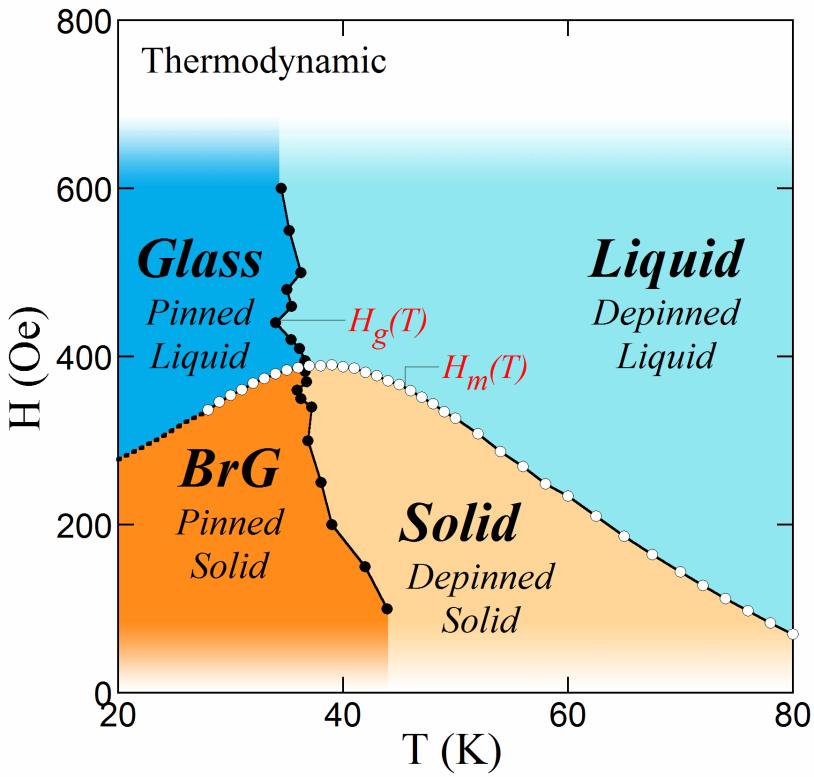
Phase Diagram Thermodynamics See examples of co2 and r134a phase diagrams and how to use them for thermodynamic analysis. learn how to use phase diagrams to understand the behavior and stability of elements, compounds and solutions as a function of temperature, pressure and composition. Unlock the secrets of phase diagrams and discover how to interpret and apply them in various thermodynamic contexts, from simple binary systems to complex multicomponent mixtures.
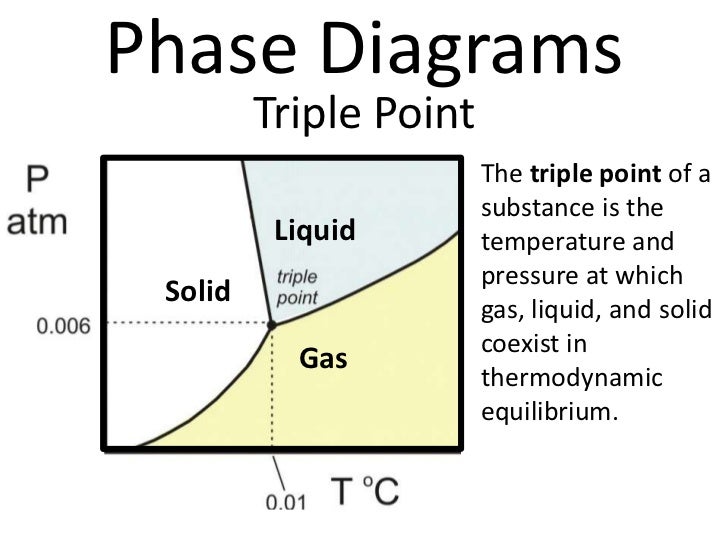
Phase Diagram Notes Flip Flop Schematic This page covers phase diagrams, critical for understanding material behaviors across states influenced by temperature, pressure, and composition. it details thermodynamic principles governing phase …. In the last lecture and leading into our discussion of phase behavior, we qualitatively described the p v t behavior of a material in the liquid vapor state, and noted that such behavior could be quantified by a cubic equation of state. We see how to use phase diagrams to judge whether two substances are mutually miscible, whether an equilibrium can exist over a range of conditions, or whether the system must be brought to a definite pressure, temperature, and composition before equilibrium can be established. In this perspective, we provide an overview of how thermodynamic phase behavior can be quantified both in computer simulations and machine learning approaches to determine phase diagrams, as well as combinations of the two.
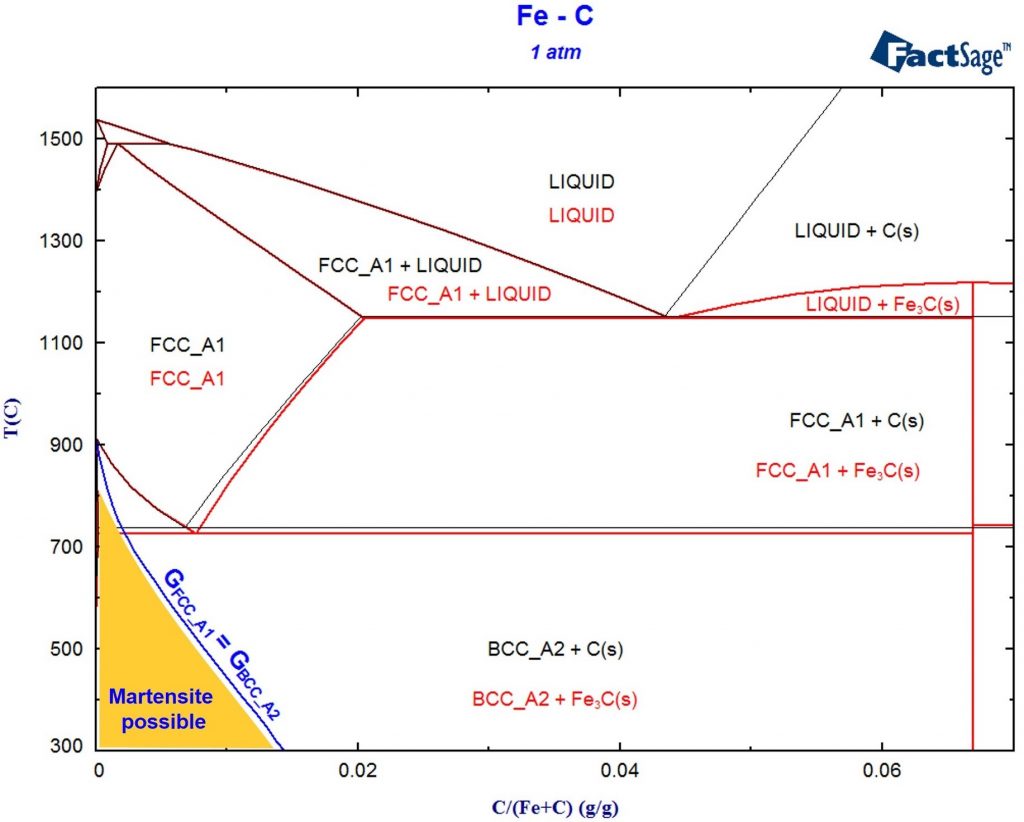
Phase Diagram Module Archives Gtt Technologies Flip Flop Schematic We see how to use phase diagrams to judge whether two substances are mutually miscible, whether an equilibrium can exist over a range of conditions, or whether the system must be brought to a definite pressure, temperature, and composition before equilibrium can be established. In this perspective, we provide an overview of how thermodynamic phase behavior can be quantified both in computer simulations and machine learning approaches to determine phase diagrams, as well as combinations of the two. Phase diagrams are graphical representations that illustrate the various phases of a substance as a function of temperature and composition, showing the conditions under which distinct phases, such as solid, liquid, and gas, exist and coexist. Standard phase diagrams are graphical representations of the equilibrium relationships between minerals (or others phases). these relationships are governed by the laws of thermodynamics. A phase diagram in physical chemistry, engineering, mineralogy, and materials science is a type of chart used to show conditions (pressure, temperature, etc.) at which thermodynamically distinct phases (such as solid, liquid or gaseous states) occur and coexist at equilibrium. When analyzing processes and cycles, these two dimensional phase diagrams are commonly used, and therefore will be discussed in detail here.

Phase Diagram Thermodynamics At Darcy Bromley Blog Phase diagrams are graphical representations that illustrate the various phases of a substance as a function of temperature and composition, showing the conditions under which distinct phases, such as solid, liquid, and gas, exist and coexist. Standard phase diagrams are graphical representations of the equilibrium relationships between minerals (or others phases). these relationships are governed by the laws of thermodynamics. A phase diagram in physical chemistry, engineering, mineralogy, and materials science is a type of chart used to show conditions (pressure, temperature, etc.) at which thermodynamically distinct phases (such as solid, liquid or gaseous states) occur and coexist at equilibrium. When analyzing processes and cycles, these two dimensional phase diagrams are commonly used, and therefore will be discussed in detail here.
Comments are closed.