Pharmaceutical Drug Development Program Implementation And Oversight Pdg

Pharmaceutical Drug Development Program Implementation And Oversight Pdg Pdg’s experience in execution and oversight of product development includes clinical studies (phases i iv), toxicology programs, formulation and cmc related assignments, good manufacturing practices (gmp), statistical assignments, product labeling and project management. Pdg navigates the complexities of the us pharmaceutical industry for clients through our strategic drug development services that include analysis of commercial potential during early stages of clinical development in parallel with regulatory planning.

Drug Development Approval Process Kuliah Pdf Phases Of Clinical This guidance describes internationally accepted principles and practices in the design and conduct of clinical studies of drug and biological products. Clinical development program are information on clinical trials that have been, or will be conducted, including any rational implementation of each study and the expected results, proving the safety and efficacy of opb required to obtain a circulation license in indonesia. These principles are applicable to all medical product development programs regardless of technology, therapeutic area, or indication, and can be applied to the development of devices and diagnostics as well. Years of service to the pharmaceutical industry has taught pdg that supplier vendor selection, audit, qualification, contract negotiation, and oversight are critical to a successful drug development program.

Drug Development Teams And Regulatory Requirements Pdf Drug These principles are applicable to all medical product development programs regardless of technology, therapeutic area, or indication, and can be applied to the development of devices and diagnostics as well. Years of service to the pharmaceutical industry has taught pdg that supplier vendor selection, audit, qualification, contract negotiation, and oversight are critical to a successful drug development program. Abstract the food and drug administration (fda) is responsible for the regulation of the pharmaceutical industry in the interest of protecting public health. the aim of this review was to outline the evolution and current role of the fda in the development and approval of new drugs. The workshop highlighted current guidances relevant to the field of clinical pharmacology, discussed the challenges and gaps in applying clinical pharmacology principles during drug development, and identified potential opportunities for further regulatory research and best practice development. Pdg offers generic pharmaceutical drug product development services to meet our clients’ regulatory needs, addressing both pre and post approval preparedness concerns. The european medicines agency's scientific guidelines on pharmaceutical development help medicine developers prepare marketing authorisation applications for human medicines. for a complete list of scientific guidelines currently open for consultation, see public consultations.
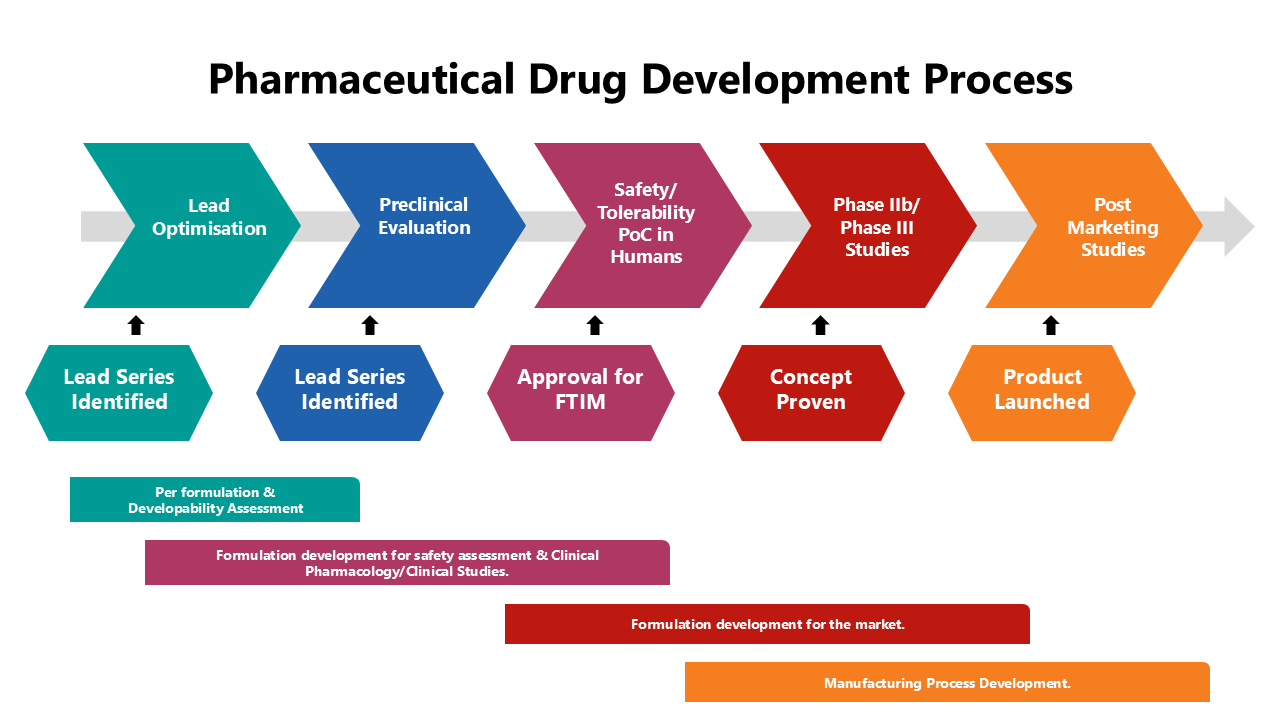
Pharmaceutical Drug Development Process Presentation Abstract the food and drug administration (fda) is responsible for the regulation of the pharmaceutical industry in the interest of protecting public health. the aim of this review was to outline the evolution and current role of the fda in the development and approval of new drugs. The workshop highlighted current guidances relevant to the field of clinical pharmacology, discussed the challenges and gaps in applying clinical pharmacology principles during drug development, and identified potential opportunities for further regulatory research and best practice development. Pdg offers generic pharmaceutical drug product development services to meet our clients’ regulatory needs, addressing both pre and post approval preparedness concerns. The european medicines agency's scientific guidelines on pharmaceutical development help medicine developers prepare marketing authorisation applications for human medicines. for a complete list of scientific guidelines currently open for consultation, see public consultations.
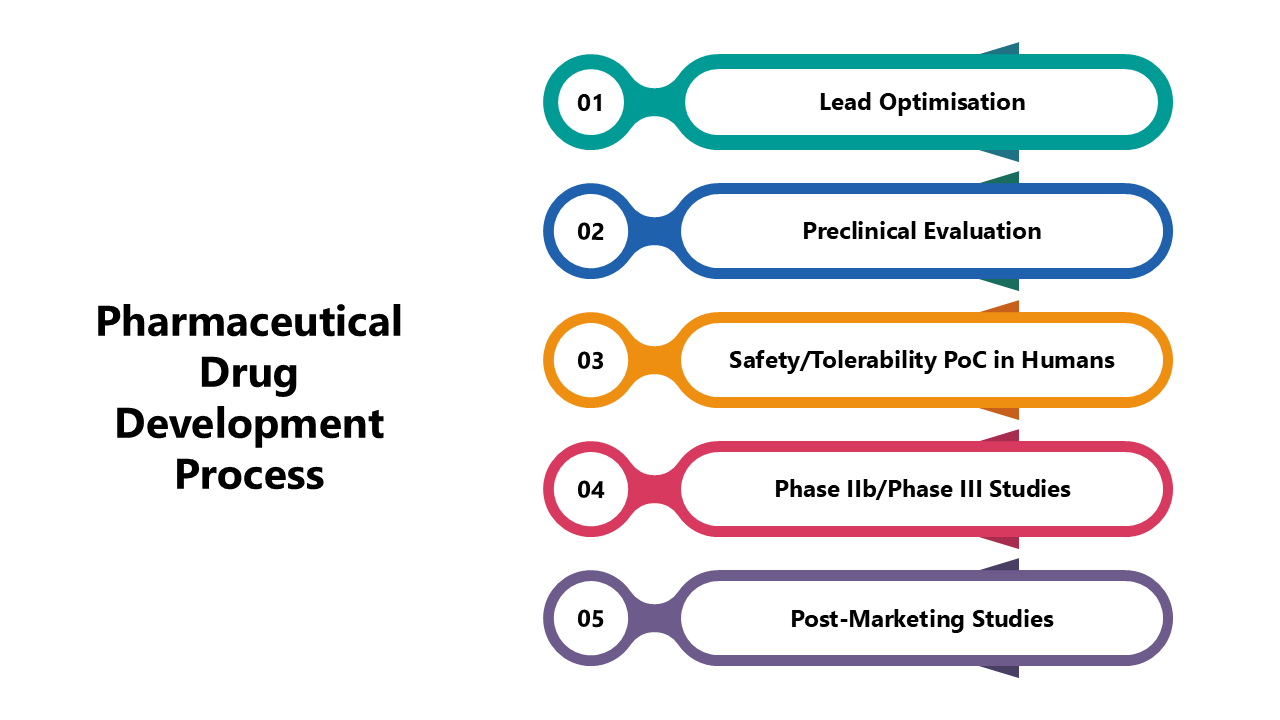
Pharmaceutical Drug Development Process Presentation Pdg offers generic pharmaceutical drug product development services to meet our clients’ regulatory needs, addressing both pre and post approval preparedness concerns. The european medicines agency's scientific guidelines on pharmaceutical development help medicine developers prepare marketing authorisation applications for human medicines. for a complete list of scientific guidelines currently open for consultation, see public consultations.
Comments are closed.