Pharma Digital Twin

Pharma Digital Twin In this review, we explore different applications of digital twins across the pharmaceutical value chain, from target discovery and preclinical research to clinical trials, regulatory review, manufacturing and post market clinical practice. Digital twins (dts) represent a groundbreaking development tool in the pharmaceutical and biopharmaceutical industries, providing virtual representations of physical entities, processes, or systems.
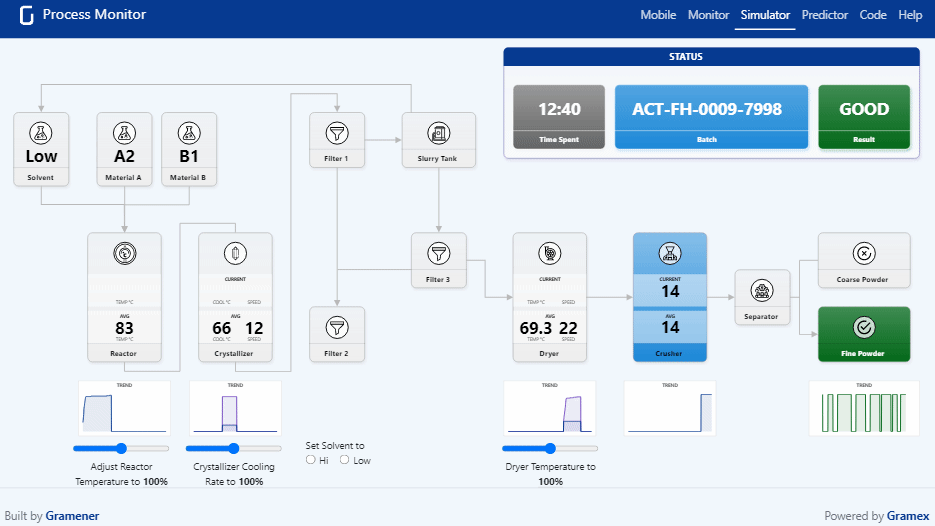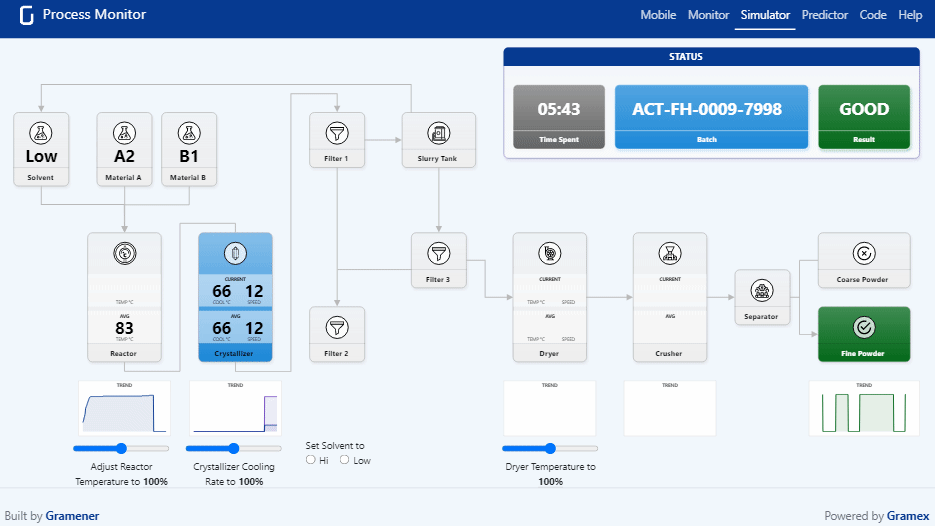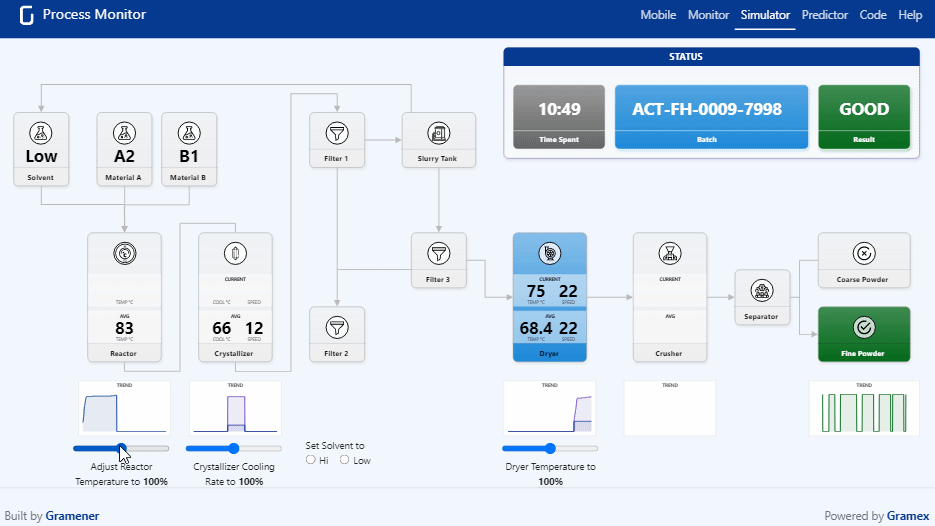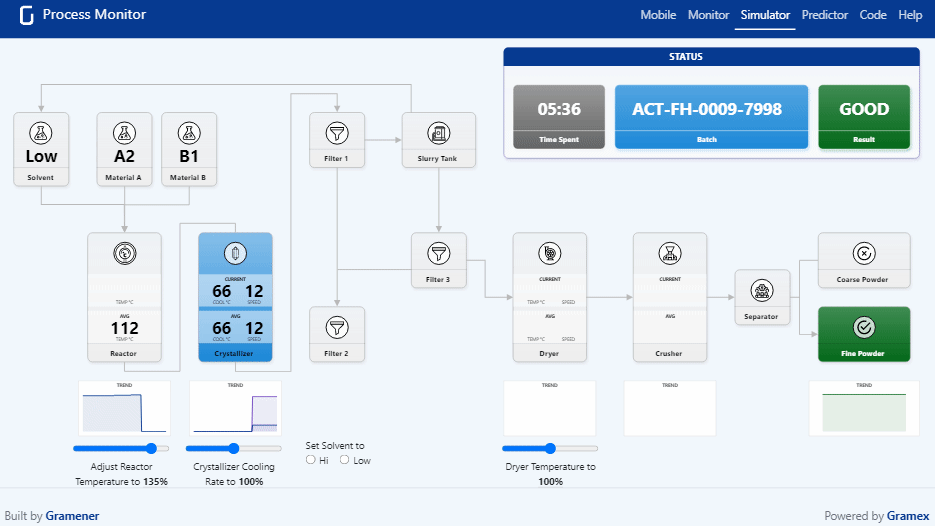
Pharma Digital Twin What is a digital twin in pharmaceutical manufacturing? a digital twin is a dynamic, virtual model of equipment, processes, or entire facilities that continuously updates using real time operational data. We present a novel, holistic approach to utilizing digital twins (dts) and ai agents throughout the pharmaceutical product lifecycle by focusing on cbps that influence both product and company performance. Digital twins have become an essential tool for pharmaceutical companies operating in a data driven, patient centric environment. their strategic use fosters continuous innovation and cross functional collaboration, with models regularly refined based on feedback and evolving market conditions. Explore how digital twins are transforming healthcare and drug discovery, with success stories showcasing their impact in the biopharma industry.

Pharma Digital Twin Digital twins have become an essential tool for pharmaceutical companies operating in a data driven, patient centric environment. their strategic use fosters continuous innovation and cross functional collaboration, with models regularly refined based on feedback and evolving market conditions. Explore how digital twins are transforming healthcare and drug discovery, with success stories showcasing their impact in the biopharma industry. Abstract— digital twin (dt) technology is emerging as a transformative innovation within the pharmaceutical and biopharmaceutical sectors, revolutionizing how drugs are developed, manufactured, and personalized. Digital twins in pharma are revolutionizing drug development, bioprocessing, and personalized medicine through the development of dynamic virtual replicas of the physical system. As a digital replica of a physical asset or process, a digital twin simulates its real world counterpart. this concept, aligned with broader trends like industry 4.0, is now accelerating drug development, optimising production, and enhancing patient outcomes. Susan schniepp, regulatory compliance associates inc., discusses digital twins and ai in pharma, focusing on data integrity, human in the loop roles, and evolving machine learning risks.
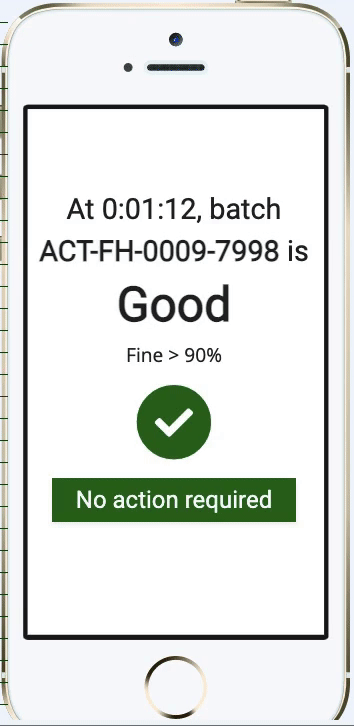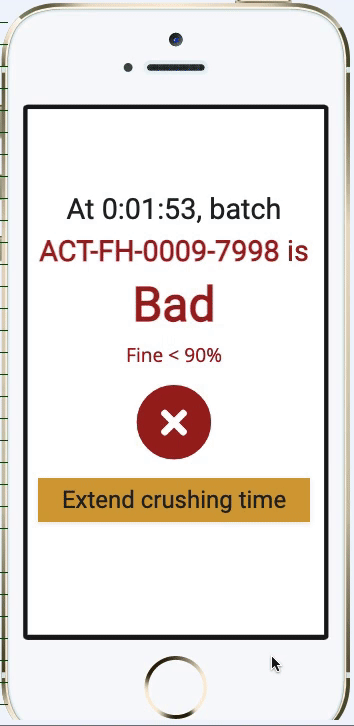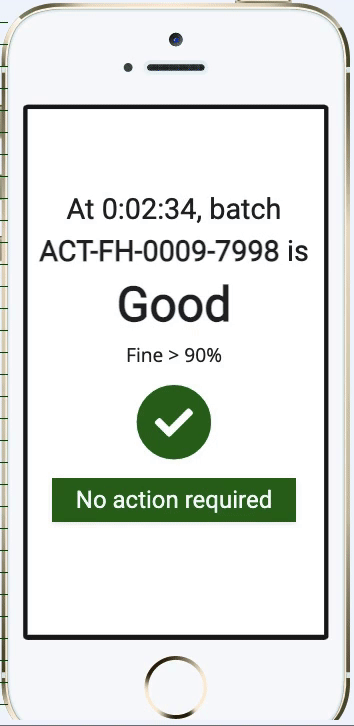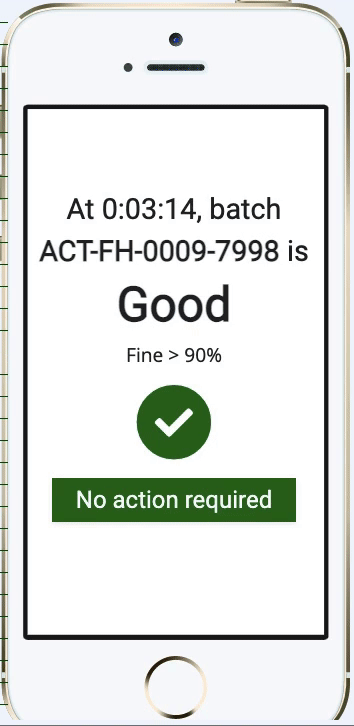
Pharma Digital Twin Abstract— digital twin (dt) technology is emerging as a transformative innovation within the pharmaceutical and biopharmaceutical sectors, revolutionizing how drugs are developed, manufactured, and personalized. Digital twins in pharma are revolutionizing drug development, bioprocessing, and personalized medicine through the development of dynamic virtual replicas of the physical system. As a digital replica of a physical asset or process, a digital twin simulates its real world counterpart. this concept, aligned with broader trends like industry 4.0, is now accelerating drug development, optimising production, and enhancing patient outcomes. Susan schniepp, regulatory compliance associates inc., discusses digital twins and ai in pharma, focusing on data integrity, human in the loop roles, and evolving machine learning risks.

Digital Twin For Pharma Industry Process Intelimek As a digital replica of a physical asset or process, a digital twin simulates its real world counterpart. this concept, aligned with broader trends like industry 4.0, is now accelerating drug development, optimising production, and enhancing patient outcomes. Susan schniepp, regulatory compliance associates inc., discusses digital twins and ai in pharma, focusing on data integrity, human in the loop roles, and evolving machine learning risks.
Comments are closed.