Periodic Table Halogens Group

Periodic Table Halogens Group Halogen, any of the six nonmetallic elements that constitute group 17 (group viia) of the periodic table. the halogen elements are fluorine (f), chlorine (cl), bromine (br), iodine (i), astatine (at), and tennessine (ts). The table below is a summary of the key physical and atomic properties of the halogens. data marked with question marks are either uncertain or are estimations partially based on periodic trends rather than observations.
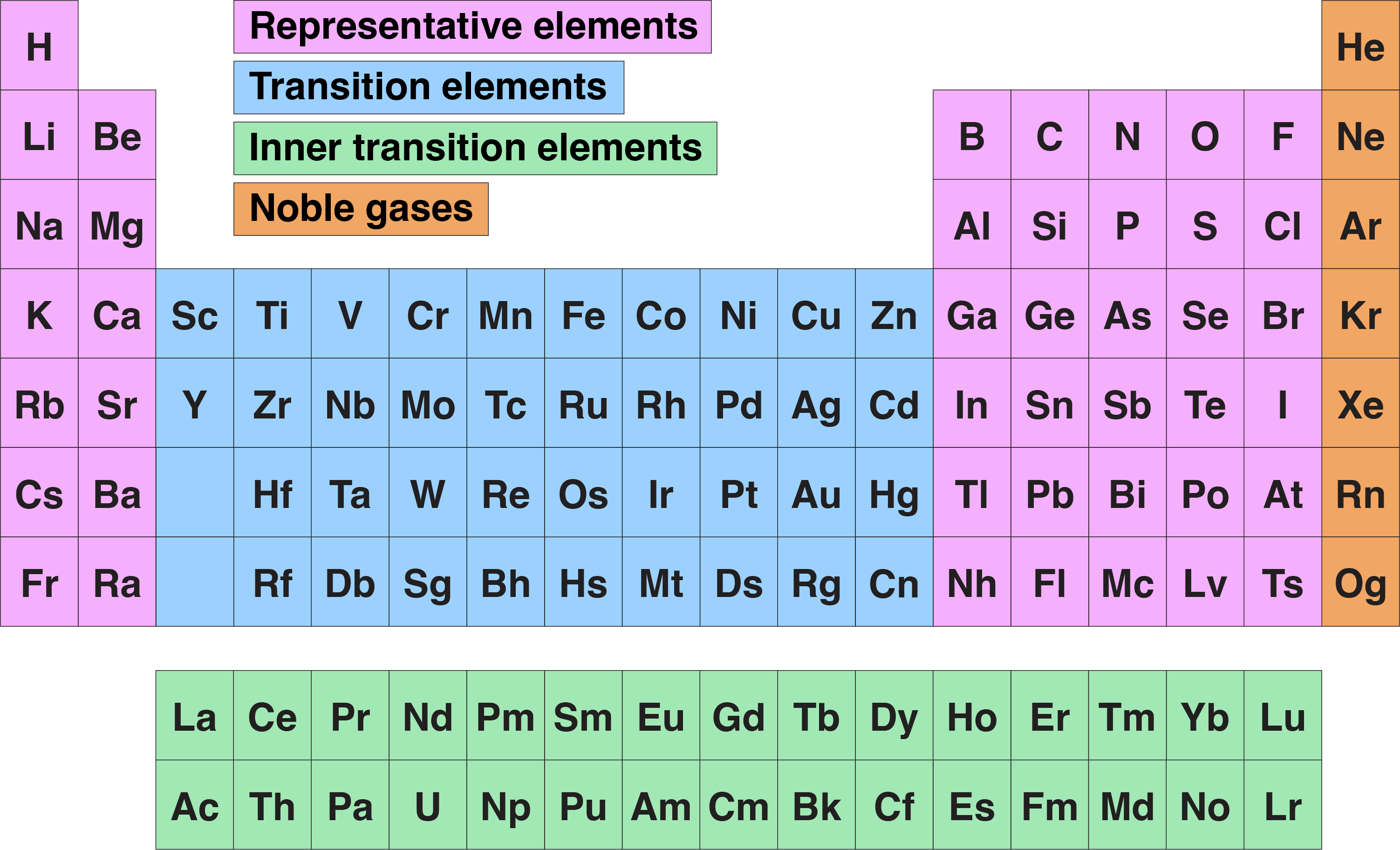
Periodic Table Halogens Group This is a list of elements that belong to the halogen group, along with information about common properties of the halogens. The halogen elements are fluorine, chlorine, bromine, iodine, astatine, and tennessine. these elements are group 17 on the periodic table. the halogens are a periodic table group of elements. they are found on the righthand side of the periodic table, just to the left of the noble gas group. The halogens are located on the left of the noble gases on the periodic table. these five toxic, non metallic elements make up group 17 of the periodic table and consist of: fluorine (f), chlorine (cl), bromine (br), iodine (i), and astatine (at). Definition: what are halogens? halogens are special category of highly reactive, non metal elements which fall in group viia of the periodic table. because of their high reactivity, the halogen family do not exist in nature in their native form, i.e. in monoatomic form.
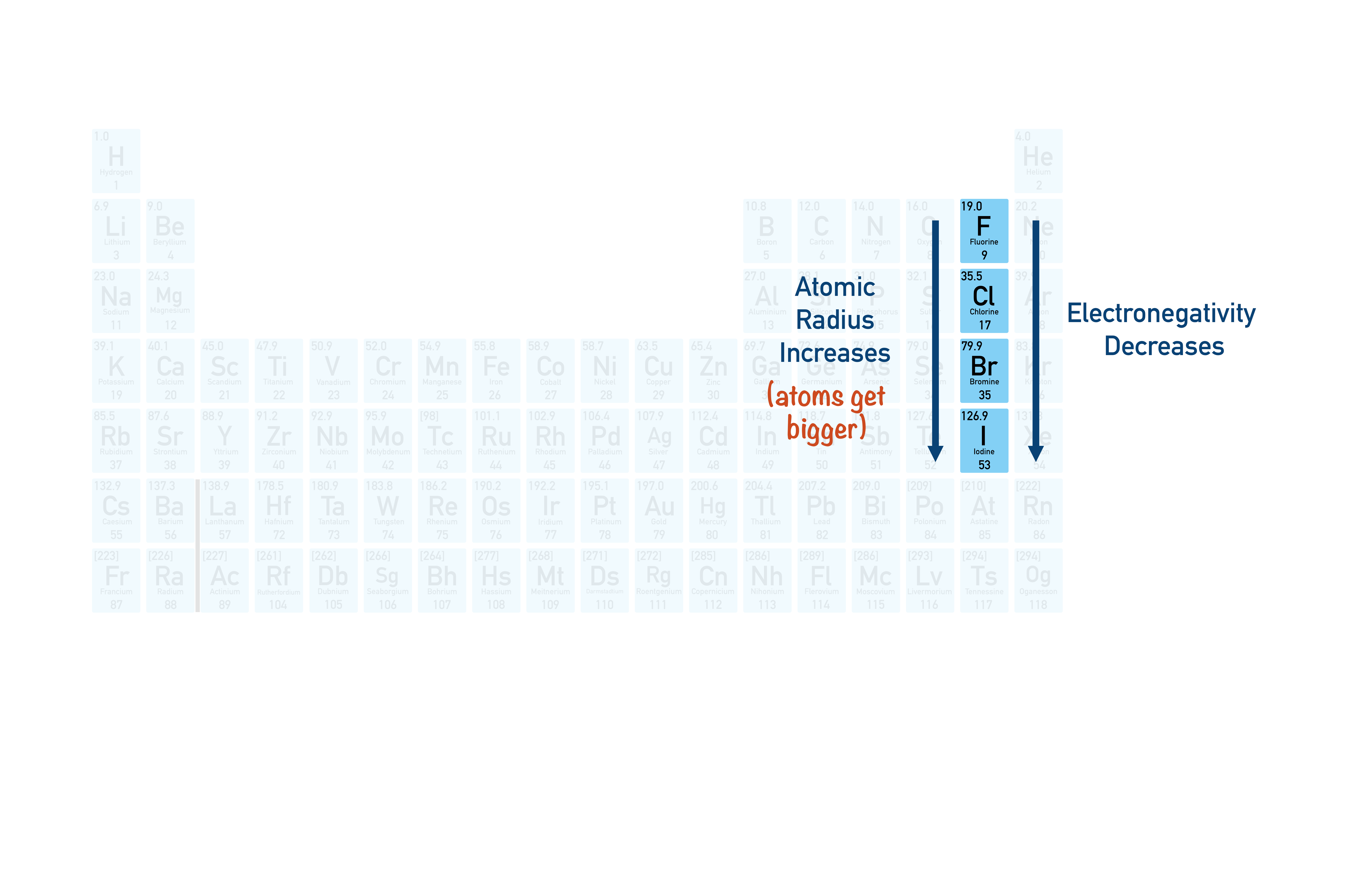
Periodic Table Halogens Group The halogens are located on the left of the noble gases on the periodic table. these five toxic, non metallic elements make up group 17 of the periodic table and consist of: fluorine (f), chlorine (cl), bromine (br), iodine (i), and astatine (at). Definition: what are halogens? halogens are special category of highly reactive, non metal elements which fall in group viia of the periodic table. because of their high reactivity, the halogen family do not exist in nature in their native form, i.e. in monoatomic form. Halogen elements are a group of highly reactive nonmetals found in group 17 (group viia) of the periodic table. "halogen" means "salt former," as these elements readily react with metals to form salts. The halogens, aka halogen family, are a group of reactive elements in group 17 of the periodic table, to the right of the chalcogens, and to the left of the noble gases. The halogens in group 17 are non metal elements that are diatomic and simple covalently bonded. they decrease in reactivity down the group but increase in their melting and boiling points. Halogen – one of the 5 non metals in group 7 of the periodic table. displacement reaction – a reaction in which a more reactive element takes the place of a less reactive element in a compound. the video below provides an overview of the halogens.
Comments are closed.