Peering Beyond The Chapter Elevating Sterile Compounding Practices

Introduction To Compounding Practices Dds Lab Mod 1 Pdf Pharmacy The applicability of the “technology statement” in usp 797, how to use official usp monographs, and the practical interpretation of "protect from light" information will increase the knowledge of. Summarize the availability and use of usp monographs for sterile compounding. using an example of sterile to sterile and api compounding, the participant will be able to identify the required tests in the usp monograph examples.

Sterile Compounding Certification Guide Qualified Join us thursday, november 14, for a fantastic webinar "peering beyond the chapter: elevating sterile compounding practices" with speaker george r. smith, pharm.d., bcps, bcscp. Course description: this course aims to improve your knowledge in the implementation of usp general chapter pharmaceutical compounding—sterile preparations. This chapter provides procedures and requirements for compounding sterile preparations. sterile compounding differs from nonsterile compounding (see pharmaceutical compounding—nonsterile preparations 795 and good compounding practices 1075) primarily by requiring a test for sterility. Beyond use dates (buds): the committee emphasized that buds are based on stability and sterility data, the compounding environment, and the financial impact on compounders and patients.

Optimizing Sterile Compounding From Aseptic Processing To Fda This chapter provides procedures and requirements for compounding sterile preparations. sterile compounding differs from nonsterile compounding (see pharmaceutical compounding—nonsterile preparations 795 and good compounding practices 1075) primarily by requiring a test for sterility. Beyond use dates (buds): the committee emphasized that buds are based on stability and sterility data, the compounding environment, and the financial impact on compounders and patients. As the leading provider of sterile and nonsterile compounding resources in the industry, we are dedicated to providing our members and customers with timely, accurate, and complete information that incorporates the new standards. Arl bio pharma posted a video "peering beyond the chapter elevating sterile compounding practices" on. This continuing education activity focuses on the essential principles, processes, and regulatory requirements of quality control and assurance in sterile compounding. Held at nhia’s annual conference, this four day, hands on experience shares the latest information and best practices for sterile compounding standards and methods, all while earning acpe approved continuing education credits.

Understanding Sterile Compounding Complimentary Webinar Life As the leading provider of sterile and nonsterile compounding resources in the industry, we are dedicated to providing our members and customers with timely, accurate, and complete information that incorporates the new standards. Arl bio pharma posted a video "peering beyond the chapter elevating sterile compounding practices" on. This continuing education activity focuses on the essential principles, processes, and regulatory requirements of quality control and assurance in sterile compounding. Held at nhia’s annual conference, this four day, hands on experience shares the latest information and best practices for sterile compounding standards and methods, all while earning acpe approved continuing education credits.
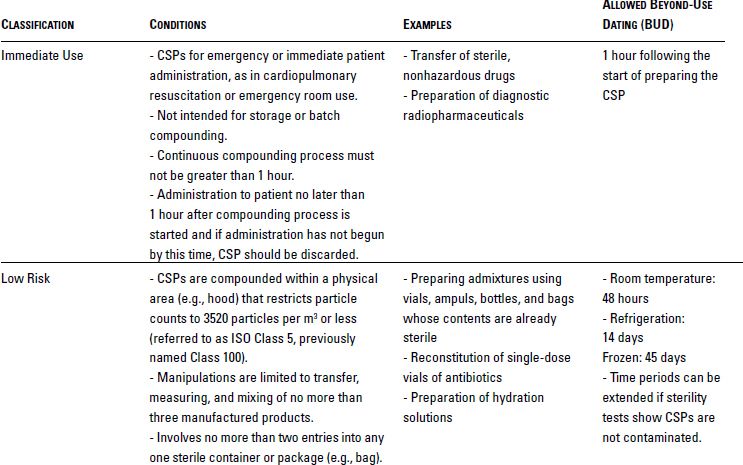
Sterile Compounding Basicmedical Key This continuing education activity focuses on the essential principles, processes, and regulatory requirements of quality control and assurance in sterile compounding. Held at nhia’s annual conference, this four day, hands on experience shares the latest information and best practices for sterile compounding standards and methods, all while earning acpe approved continuing education credits.
Comments are closed.