Pdf Using Real World Data Alongside Evidence In Guideline Development

Real World Evidence And Real World Data Pdf This article summarizes the nice process for using real world evidence in clinical guidelines which can be adapted to the ihope context. Pdf | on may 12, 2022, anirudh gaurang gudlavalleti and others published using real world data alongside evidence in guideline development | find, read and cite all the research.

Pdf Translating Real World Evidence Real World Data Semantic scholar extracted view of "using real world data alongside evidence in guideline development" by a. g. gudlavalleti et al. Ongoing development of methodologies around real world data has improved our understanding of what constitutes real world evidence and in what circumstances it is expected to be most reliable and useful. Your smart literature assistant to help you manage science literatures, read pdf, take notes, cite literature in word. This paper provides a high level summary of this report. more work remains to be done to globally harmonize practices and guidance for using rwd and rwe for regulatory decision making, thereby maximizing the benefits they can bring to patient care and public health.

Fda S Guidelines On Use Of Real World Data Omny Health Your smart literature assistant to help you manage science literatures, read pdf, take notes, cite literature in word. This paper provides a high level summary of this report. more work remains to be done to globally harmonize practices and guidance for using rwd and rwe for regulatory decision making, thereby maximizing the benefits they can bring to patient care and public health. 110 this proposal will aim to benefit all types of medicinal products at any stage of their lifecycle, i.e., from 111 development pre approval to post marketing monitoring. ich guidance can increase the efficiency of 112 resources across a large number of stakeholders, by aligning expectations of medicines regulators, 113 medicines developers. Pursuant to this section, fda created a framework for a program (rwe program) to evaluate the potential use of real world evidence (rwe) in regulatory decision making. The main tools for the transparency of nis are the ema hma catalogue of 386 data sources (described in the good practice guide for the use of the metadata catalogue of real 387 world data sources) and the ema hma catalogue of non interventional studies (14). Rwd enables real world evidence development to complement and supplement randomized controlled trials (rcts). rwd can be translated to rwe to provide insights into treatment effectiveness, safety and outcomes beyond controlled clinical trials.

Pdf Improving The Use Of Research Evidence In Guideline Development 110 this proposal will aim to benefit all types of medicinal products at any stage of their lifecycle, i.e., from 111 development pre approval to post marketing monitoring. ich guidance can increase the efficiency of 112 resources across a large number of stakeholders, by aligning expectations of medicines regulators, 113 medicines developers. Pursuant to this section, fda created a framework for a program (rwe program) to evaluate the potential use of real world evidence (rwe) in regulatory decision making. The main tools for the transparency of nis are the ema hma catalogue of 386 data sources (described in the good practice guide for the use of the metadata catalogue of real 387 world data sources) and the ema hma catalogue of non interventional studies (14). Rwd enables real world evidence development to complement and supplement randomized controlled trials (rcts). rwd can be translated to rwe to provide insights into treatment effectiveness, safety and outcomes beyond controlled clinical trials.
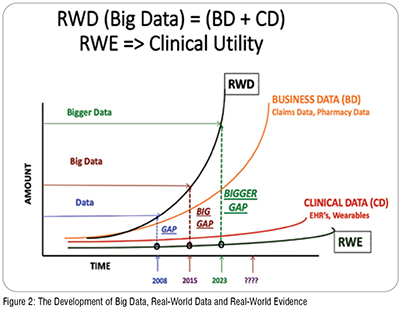
Real World Evidence In Healthcare Research Development Pharma The main tools for the transparency of nis are the ema hma catalogue of 386 data sources (described in the good practice guide for the use of the metadata catalogue of real 387 world data sources) and the ema hma catalogue of non interventional studies (14). Rwd enables real world evidence development to complement and supplement randomized controlled trials (rcts). rwd can be translated to rwe to provide insights into treatment effectiveness, safety and outcomes beyond controlled clinical trials.

Pdf A Path To Real World Evidence In Critical Care Using Open Source
Comments are closed.