Pdf A Randomized Placebo Controlled Double Blinded Parallel Group

Chandrasekhar Et Al 2012 A Prospective Randomized Double Blind Placebo The design of the clinical trial is described in fig. 1 and is a phase iia randomised, double blind, placebo controlled, parallel group, and multi centric trial, carried out between. Pdf | objective: alpha brain® is a nootropic supplement that purports to enhance cognitive functioning in healthy adults.
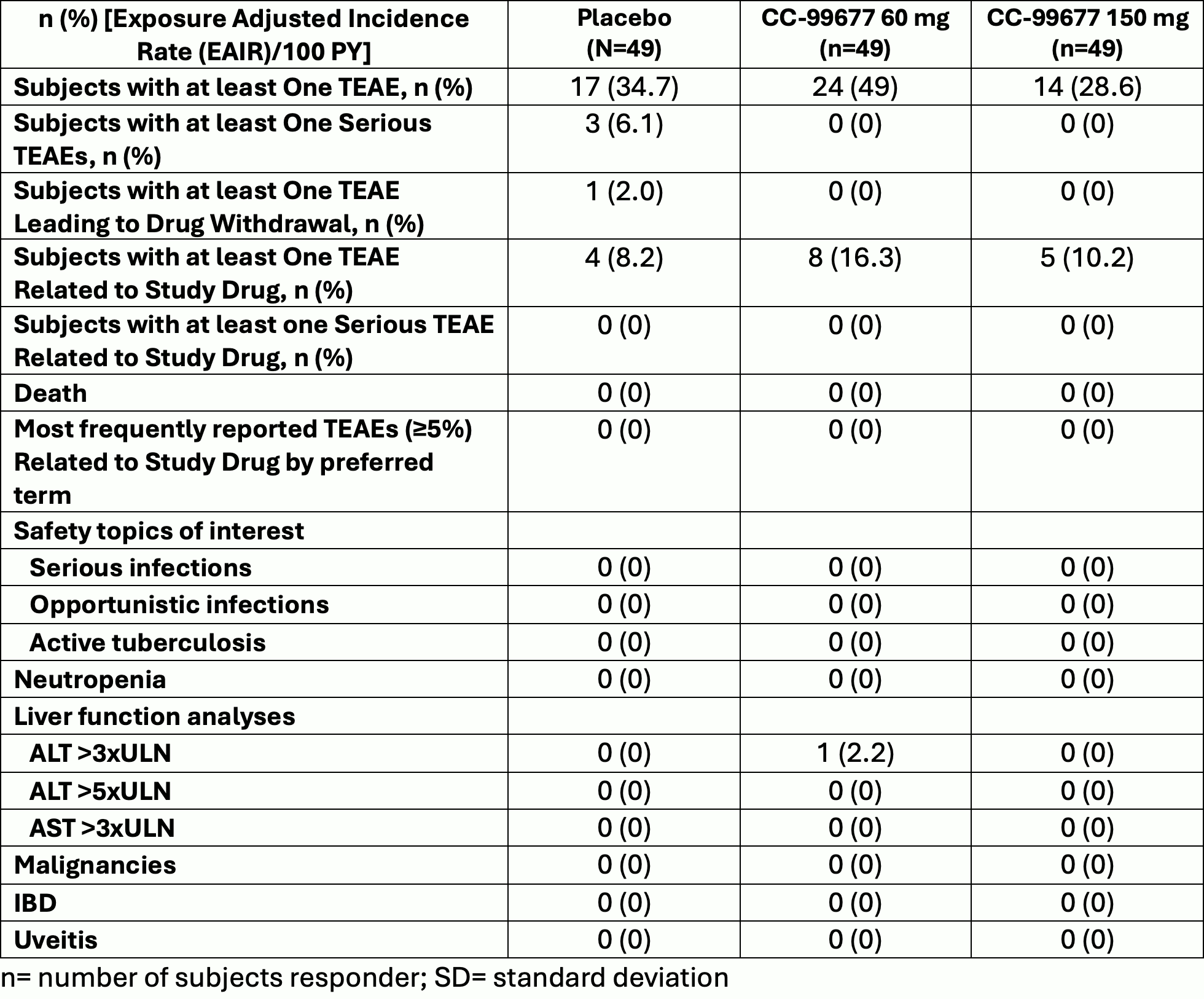
A Phase 2 Multicenter Randomized Double Blinded Placebo Controlled We evaluated the efficacy and safety of an mts 9 hour wear time in comparison with placebo and, with reference to an established adhd treat ment (oros methylphenidate), in a 7 week, randomized, placebo controlled trial with > 200 pediatric participants diagnosed with adhd. The sonata hcm is a phase 3, randomized, double blind, placebo controlled multicenter trial that will evaluate efficacy of sotagliflozin, a dual sglt1 and sglt2 inhibitor, on symptoms, function, and other patient reported outcomes (pros), as well as safety in patients with symptomatic hcm. Ruby 1: a randomized, double blind, placebo controlled trial of the safety and tolerability of the nov el oral factor xa inhibitor darexaban (ym150) following acute coronary syn drome. Randomized double blind placebo control (rdbpc) studies are considered the “gold standard” of epidemiologic studies.
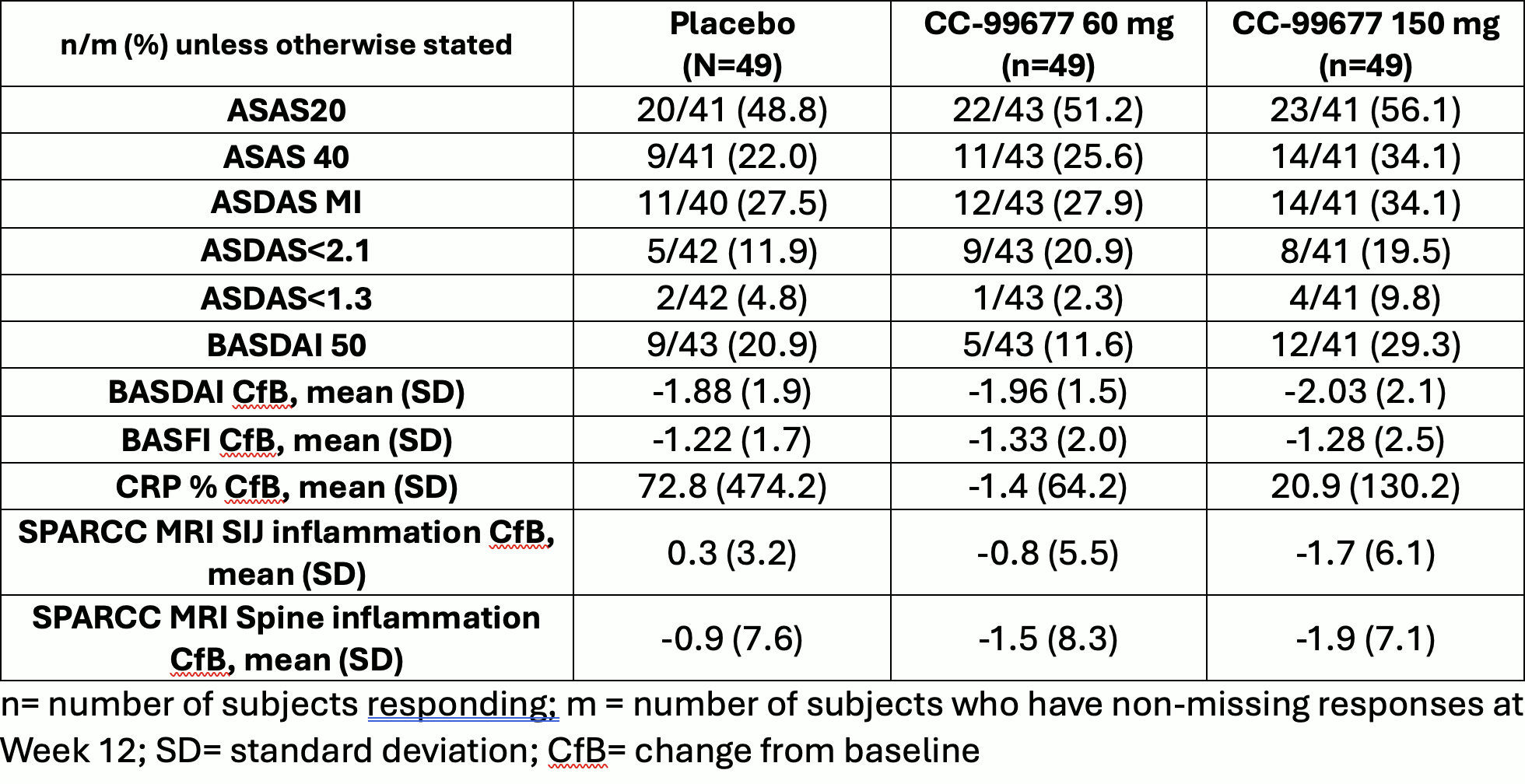
A Phase 2 Multicenter Randomized Double Blinded Placebo Controlled Ruby 1: a randomized, double blind, placebo controlled trial of the safety and tolerability of the nov el oral factor xa inhibitor darexaban (ym150) following acute coronary syn drome. Randomized double blind placebo control (rdbpc) studies are considered the “gold standard” of epidemiologic studies. Study alxn1210 als 308 is a phase 3, double blind, randomized, placebo controlled, parallel group, multicenter study with an ole to evaluate the efficacy and safety of ravulizumab in adult patients with amyotrophic lateral sclerosis (als). A phase 2a, randomized, double blind, placebo controlled, three arm, parallel group study to assess the efficacy, safety, tolerability and pharmacodynamics of pf 06835919 in patients with non alcoholic fatty liver disease and type 2 diabetes. Cascade is an ongoing exploratory, phase 2, random ized, double blind, placebo controlled, parallel group study that aims to assess the anti inflammatory effects of tezepelumab 210 mg administered subcutaneously every 4 weeks (q4w) for 28 weeks in adults (aged 18 75 years). This is a randomized, double blinded, placebo controlled, parallel group, multicenter, operationally seamless phase 2 3 study to evaluate the efficacy, safety, tolerability, pk, pd, and immunogenicity of efgartigimod ph20 sc in adult participants with active iim.

A Multicenter Randomized Double Blinded Parallel Group Placebo Study alxn1210 als 308 is a phase 3, double blind, randomized, placebo controlled, parallel group, multicenter study with an ole to evaluate the efficacy and safety of ravulizumab in adult patients with amyotrophic lateral sclerosis (als). A phase 2a, randomized, double blind, placebo controlled, three arm, parallel group study to assess the efficacy, safety, tolerability and pharmacodynamics of pf 06835919 in patients with non alcoholic fatty liver disease and type 2 diabetes. Cascade is an ongoing exploratory, phase 2, random ized, double blind, placebo controlled, parallel group study that aims to assess the anti inflammatory effects of tezepelumab 210 mg administered subcutaneously every 4 weeks (q4w) for 28 weeks in adults (aged 18 75 years). This is a randomized, double blinded, placebo controlled, parallel group, multicenter, operationally seamless phase 2 3 study to evaluate the efficacy, safety, tolerability, pk, pd, and immunogenicity of efgartigimod ph20 sc in adult participants with active iim.
Comments are closed.