Pauli Exclusion Principle Statement Examples Importance

Pauli Exclusion Principle Formulas Equations Differents Faqs What does the pauli exclusion principle state. learn its definition and applications, along with examples and illustrations. The pauli exclusion principle dictates that the presence of two electrons in a given material occupying the same energy state is prohibited. the comprehension of fermi levels within the solid state band theory can be enhanced through the utilization of pauli’s exclusion principle.

Pauli Exclusion Principle Statement Examples Importance According to pauli’s exclusion principle, “no two electrons in an atom can have the same set of all four quantum numbers”. only two electrons are allowed to share an orbital, and they must have the opposite spin. as an illustration of pauli's exclusion principle, we use a neutral helium atom. An austrian theoretical physicist wolfgang ernst pauli in 1925 put forward an ingenious principle known as pauli exclusion principle. this fundamental principle controls the assignment of values of four quantum numbers of an electron in an orbital. The pauli exclusion principle helps explain a wide variety of physical phenomena. one particularly important consequence of the principle is the elaborate electron shell structure of atoms and the way atoms share electrons, explaining the variety of chemical elements and their chemical combinations. The pauli exclusion principle is an important rule that explains how electrons are arranged in the orbitals of an atom. according to this principle, no two electrons in an atom can have the same set of four quantum numbers.
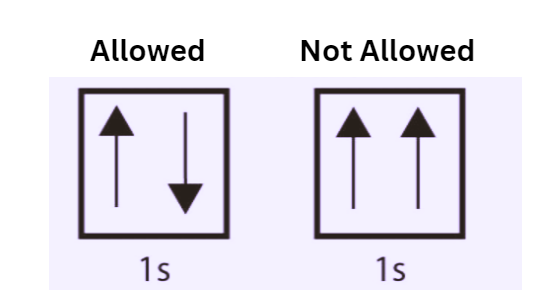
Pauli Exclusion Principle Statement Examples Importance The pauli exclusion principle helps explain a wide variety of physical phenomena. one particularly important consequence of the principle is the elaborate electron shell structure of atoms and the way atoms share electrons, explaining the variety of chemical elements and their chemical combinations. The pauli exclusion principle is an important rule that explains how electrons are arranged in the orbitals of an atom. according to this principle, no two electrons in an atom can have the same set of four quantum numbers. The pauli exclusion principle states that no two electrons in an atom can have identical quantum numbers. the pauli exclusion principle is an important concept in chemistry and in quantum mechanics in physics. One explanation as to why the differences between the term symbols that arise from a p 2 configuration relative to a pp configuration is the pauli exclusion principle. A maximum of two electrons can occupy an orbital. to obey pauli’s principle, these two electrons must differ in spin quantum number (ms = ½ or –½). example: in the 1s orbital of helium (1s²), both electrons occupy the same orbital (n=1, ℓ=0, mℓ=0), but one has ms = ½ and the other ms = –½. The pauli exclusion principle states that no two electrons in an atom can have the same set of all four quantum numbers. the principle ensures that electrons occupy different orbitals and have opposite spins within the same orbital.
Comments are closed.