Pauli Exclusion Principle No Identical Fermions States

The Pauli Exclusion Principle Fermions Ppt Template St Ai Ss Ppt Example In quantum mechanics, the pauli exclusion principle (german: pauli ausschlussprinzip) states that two or more identical particles with half integer spins (i.e. fermions) cannot simultaneously occupy the same quantum state within a system that obeys the laws of quantum mechanics. Tating that no two identical fermions can occupy the same quantum state simultaneously. this principle has not only revolutionized the theoretical framework of modern physics but also.
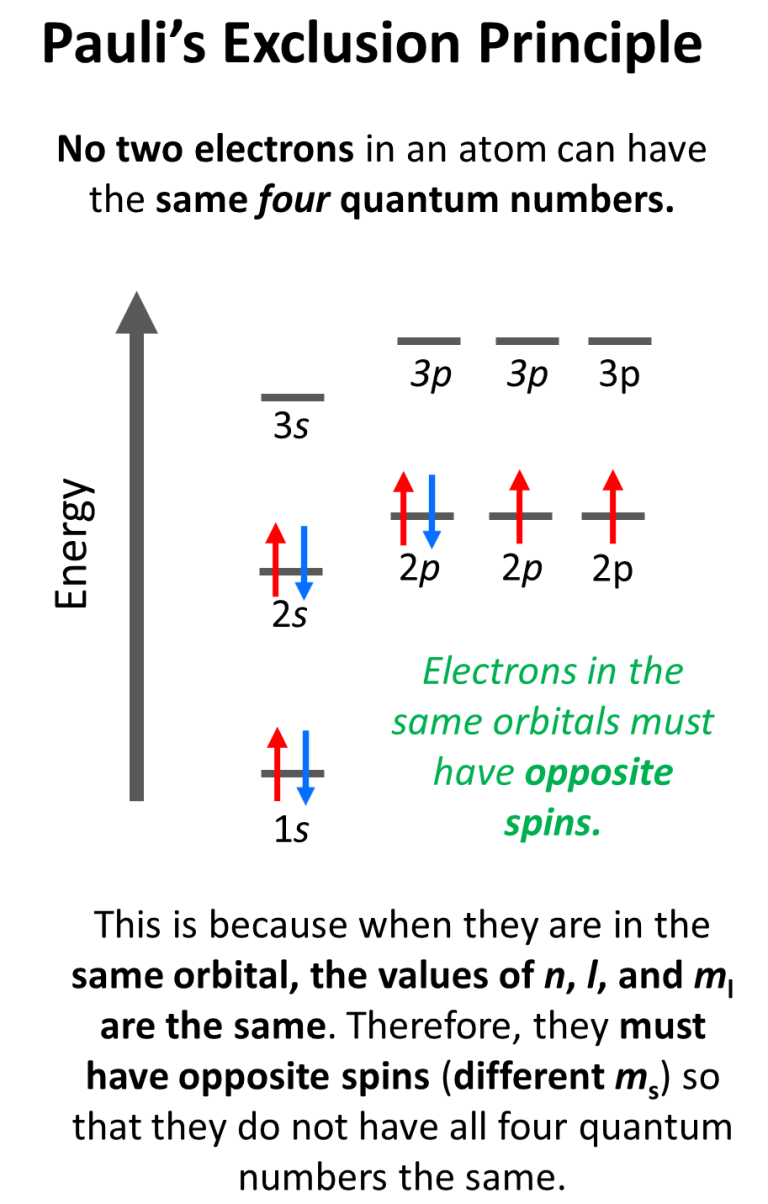
Pauli Exclusion Principle Chemistry Steps Here is the definition of the pauli exclusion principle and a simple explanation of what it means for electrons and other fermions. the pauli exclusion principle states that no two electrons or other fermions in an atom or molecule have the same electronic quantum numbers (n, l, ml, ms). The pauli exclusion principle is defined as the rule stating that no two electrons in the same atom can have identical sets of all four quantum numbers; thus, if two electrons occupy the same orbital, they must have opposite spin quantum numbers. The pauli exclusion principle is the quantum mechanical principle that states that two or more identical fermions (particles with half integer spin) cannot occupy the same quantum state within a quantum system simultaneously. The pauli exclusion principle states that no two fermions may occupy the same quantum state. this principle is absolutely crucial to life as we know it; without it, we would not have the periodic table of chemistry, nor would we have a lot of the rest of the structure of matter.

Solved Pauli Exclusion Principle Applies To A ï All Fermions Chegg The pauli exclusion principle is the quantum mechanical principle that states that two or more identical fermions (particles with half integer spin) cannot occupy the same quantum state within a quantum system simultaneously. The pauli exclusion principle states that no two fermions may occupy the same quantum state. this principle is absolutely crucial to life as we know it; without it, we would not have the periodic table of chemistry, nor would we have a lot of the rest of the structure of matter. What is the pauli exclusion principle? simply put, the pauli exclusion principle states that no two fermions (particles like electrons, protons, and neutrons that have half integer spin) can occupy the same quantum state simultaneously within a quantum system. Pauli exclusion principle, assertion that no two electrons in an atom can be at the same time in the same state or configuration, proposed (1925) by the austrian physicist wolfgang pauli to account for the observed patterns of light emission from atoms. This paper is devoted to one of the fundamental principles of quantum mechanics—the pauli exclusion principle (pep). since the formulation of pep was not derived from general concepts of quantum mechanics and based on experimental data, its theoretical foundation is still not based rigorously. The pauli exclusion principle, formulated by wolfgang pauli in 1925, states that no two fermions (such as electrons, protons, or neutrons) can occupy the same quantum state within a quantum system simultaneously.
Comments are closed.