P Block Elements Groups 13 To 18 Classification Electronic

Block Classification Of Periodic Table Elements Periods And Groups P block elements (groups 13–18) class 11 notes with electronic configuration, properties, trends & faqs for jee, neet, and cbse exam preparation. Detailed notes on p block elements group 13 to 18 for jee main and advanced. covers trends, properties, reactions, compounds with equations.

P Block Elements Fun Science P block elements group 13 to 18 ncert as per new syllabus jee main 2025 free download as pdf file (.pdf) or read online for free. Learn about the p block elements in chemistry. discover which elements they are, their location, the electron configuration, and properties. P block elements are chemical elements in which the last electron enters the p orbital of their atomic structure. these elements are found in groups 13 to 18 of the periodic table and include a diverse range of nonmetals, metalloids, and noble gases. Elements are grouped into blocks based on the type of orbital receiving the last electron. s block: groups 1 and 2, have ns 1 or ns 2 configuration, highly reactive metals. p block: groups 13–18, configurations end in np 1 to np 6, includes metals, non metals, and metalloids.
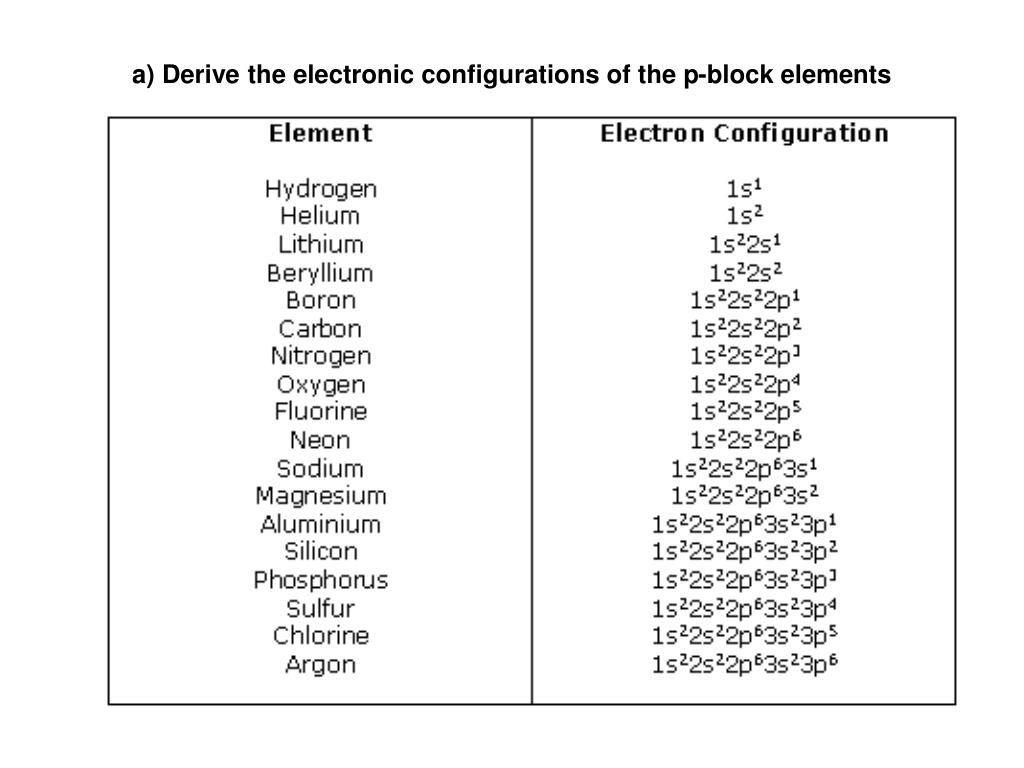
Ppt A Derive The Electronic Configurations Of The P Block Elements P block elements are chemical elements in which the last electron enters the p orbital of their atomic structure. these elements are found in groups 13 to 18 of the periodic table and include a diverse range of nonmetals, metalloids, and noble gases. Elements are grouped into blocks based on the type of orbital receiving the last electron. s block: groups 1 and 2, have ns 1 or ns 2 configuration, highly reactive metals. p block: groups 13–18, configurations end in np 1 to np 6, includes metals, non metals, and metalloids. P block elements are those elements found in groups 13 to 18 of the periodic table. these elements have their outermost electrons in the p orbital. the p block contains a diverse range of elements, including metals, non metals, and metalloids, with properties varying widely across the block. P block elements are those elements in which the last electron enters the p orbital. they occupy groups 13 18 of the periodic table and include metals, metalloids, and non metals. The modern periodic table is divided into 4 different blocks s block, p block, d block and f block. elements belonging to group 13 to group 18 are classified as p block elements because the last electron for all the elements enters into p orbitals. This lesson explores the properties, classification, and trends of p block elements in the periodic table for cbse class 11 (aligned with the ncert textbook). you will learn about their electronic configurations, oxidation states, bonding nature, and periodic trends.
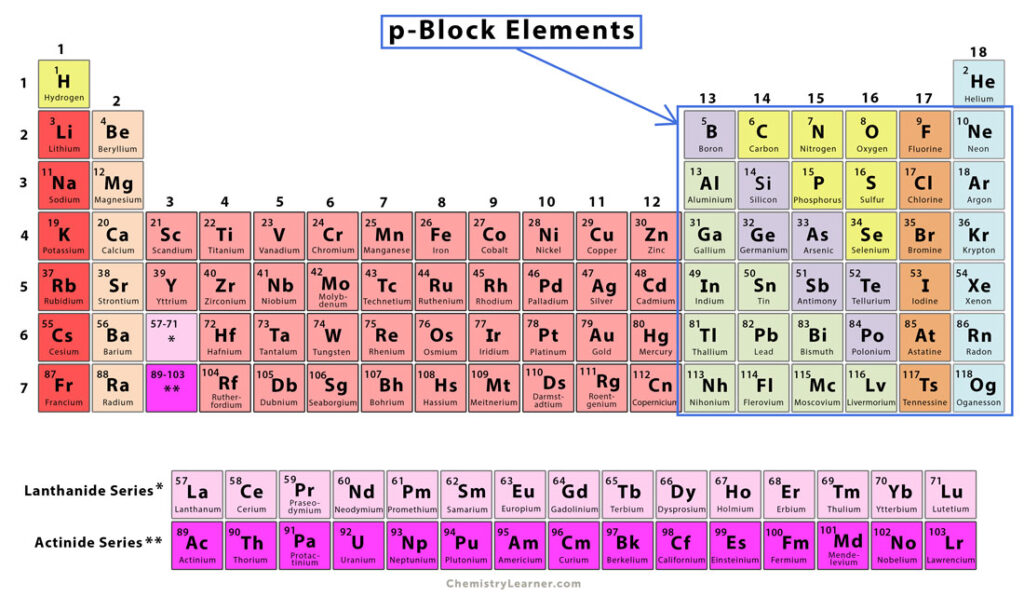
P Block Elements Chemistry Learner P block elements are those elements found in groups 13 to 18 of the periodic table. these elements have their outermost electrons in the p orbital. the p block contains a diverse range of elements, including metals, non metals, and metalloids, with properties varying widely across the block. P block elements are those elements in which the last electron enters the p orbital. they occupy groups 13 18 of the periodic table and include metals, metalloids, and non metals. The modern periodic table is divided into 4 different blocks s block, p block, d block and f block. elements belonging to group 13 to group 18 are classified as p block elements because the last electron for all the elements enters into p orbitals. This lesson explores the properties, classification, and trends of p block elements in the periodic table for cbse class 11 (aligned with the ncert textbook). you will learn about their electronic configurations, oxidation states, bonding nature, and periodic trends.
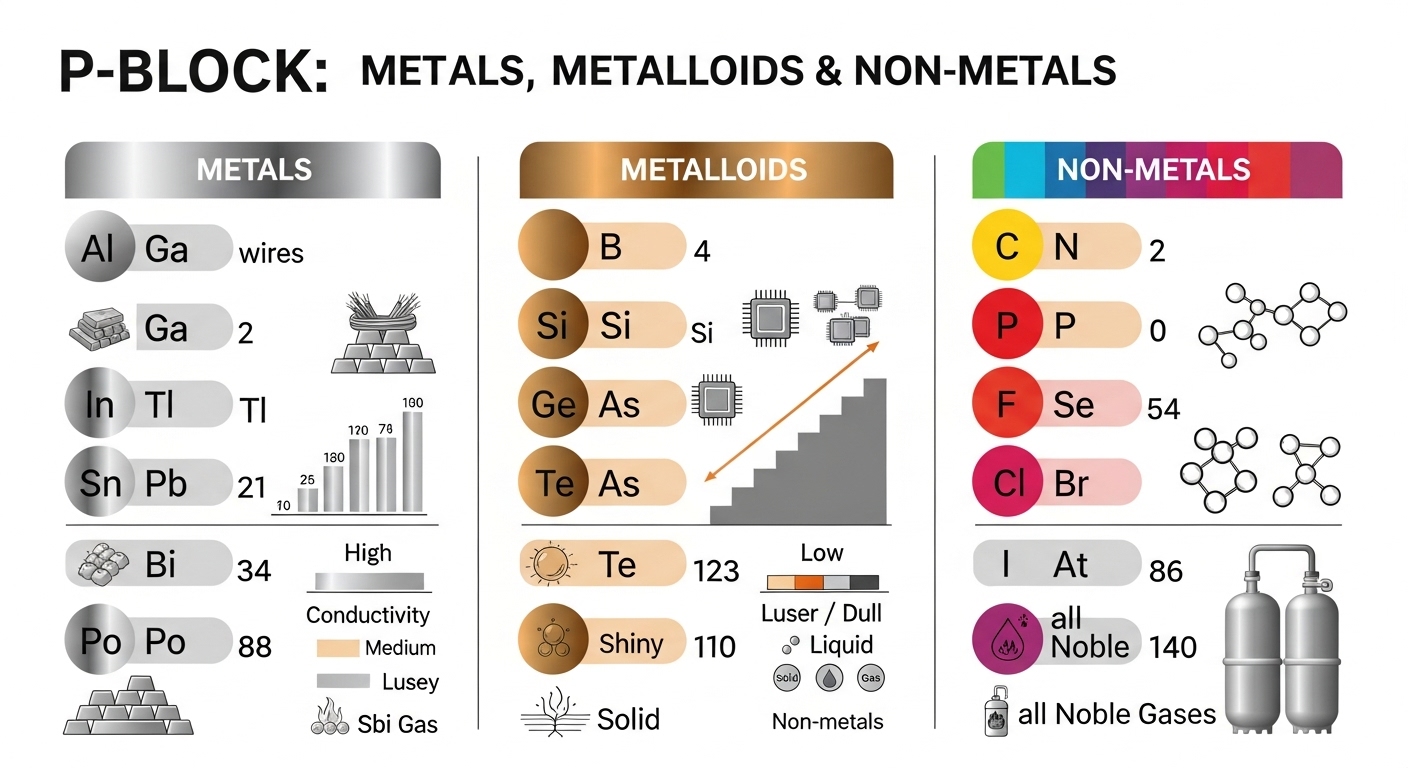
The P Block Elements Complete Guide For Class 12 Chemistry Groups 13 The modern periodic table is divided into 4 different blocks s block, p block, d block and f block. elements belonging to group 13 to group 18 are classified as p block elements because the last electron for all the elements enters into p orbitals. This lesson explores the properties, classification, and trends of p block elements in the periodic table for cbse class 11 (aligned with the ncert textbook). you will learn about their electronic configurations, oxidation states, bonding nature, and periodic trends.

Understanding P Block Elements In The Periodic Table
Comments are closed.