Oxidation And Reduction Chemash
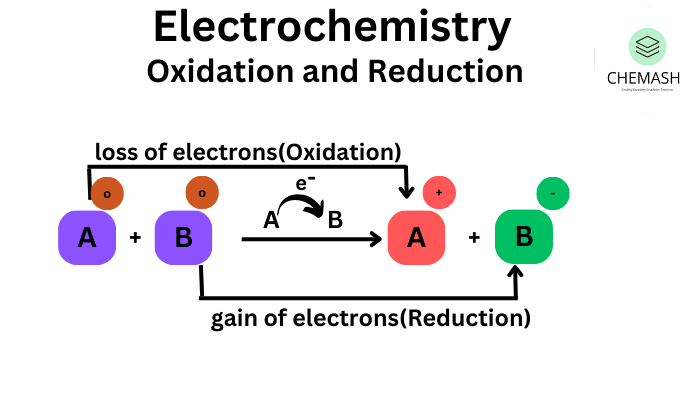
Oxidation And Reduction Chemash Learn oxidation and reduction (redox) with definitions, examples, daily life applications, mcqs, a short quiz, and faqs. This page discusses the various definitions of oxidation and reduction (redox) in terms of the transfer of oxygen, hydrogen, and electrons. it also explains the terms oxidizing agent and reducing ….

Oxidation Reduction Biochemistry This tutorial covers oxidation reduction (redox) reactions in electrochemistry: learn to write simple half reactions and to recognize the oxidizing agent and reducing agent. What is oxidation and reduction in chemistry? oxidation and reduction are types of chemical reactions where substances exchange electrons. in oxidation, a substance loses electrons (or gains oxygen), while in reduction, a substance gains electrons (or loses oxygen). Oxidation–reduction (redox) reactions (article) | khan academy khan academy. The term oxidation was originally used to describe chemical reactions involving o 2, but its meaning has evolved to refer to a broad and important reaction class known as oxidation reduction (redox) reactions.
Types Of Electrodes Chemash Oxidation–reduction (redox) reactions (article) | khan academy khan academy. The term oxidation was originally used to describe chemical reactions involving o 2, but its meaning has evolved to refer to a broad and important reaction class known as oxidation reduction (redox) reactions. Oxidation reduction (redox) reactions are those in which one or more elements involved undergo a change in oxidation number. returning to the reactions used to introduce this topic, they may now both be identified as redox processes. An important feature of oxidation reduction reactions can be recognized by examining what happens to the copper in this pair of reactions. the first reaction converts copper metal into cuo, thereby transforming a reducing agent (cu) into an oxidizing agent (cuo). Oxidation and reduction reactions are generally called redox reactions. redox reactions are also used in a wide range of industries, including those in the biological, industrial, pharmaceutical, metallurgical, and agricultural fields. Oxidation reduction reactions are now defined as reactions that exhibit a change in the oxidation states of one or more elements in the reactants by a transfer of electrons, which follows the mnemonic "oxidation is loss, reduction is gain", or "oil rig".

Oxidation Reduction Oxidation reduction (redox) reactions are those in which one or more elements involved undergo a change in oxidation number. returning to the reactions used to introduce this topic, they may now both be identified as redox processes. An important feature of oxidation reduction reactions can be recognized by examining what happens to the copper in this pair of reactions. the first reaction converts copper metal into cuo, thereby transforming a reducing agent (cu) into an oxidizing agent (cuo). Oxidation and reduction reactions are generally called redox reactions. redox reactions are also used in a wide range of industries, including those in the biological, industrial, pharmaceutical, metallurgical, and agricultural fields. Oxidation reduction reactions are now defined as reactions that exhibit a change in the oxidation states of one or more elements in the reactants by a transfer of electrons, which follows the mnemonic "oxidation is loss, reduction is gain", or "oil rig".
Comments are closed.