Nitrogen Cycle Nitrogen Fixation Pptx

Nitrogen Cycle Nitrogen Fixation Pptx The nitrogen cycle involves nitrogen fixation, ammonification, nitrification, and denitrification processes carried out by microorganisms. nitrogen fixation converts atmospheric nitrogen gas into ammonium which can then be used by plants and other organisms. Species effects can be much greater than differences in climate. 1. mineralization immobilization nearly all nitrogen that is mineralized in these systems is nitrified on a net basis. in contrast, net nitrification is frequently less than 25% of net mineralization in temperate coniferous forests.
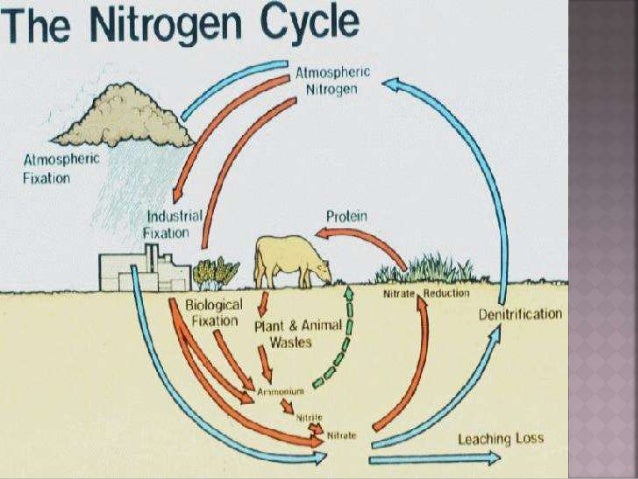
Nitrogen Fixation Cycle In order for plants and animals to be able to use nitrogen, n2 gas must first be converted to more a chemically available form such as ammonium (nh4 ) or nitrate (no3 ). this is called fixation we can’t! nitrogen fixation (n2 > nh3 or nh4 ) how?. Most of the biological systems which participate in the global nitrogen cycle contain metal requiring enzymes. three main processes can be distinguished in the nitrogen cycle: nitrogen fixation, nitrification and denitrification. n2 3h2 >400 c > 100 bar 2nh3 nitrogen fixation (industrial). Assimilation plants require nitrogen in the form of dissolved nitrates no3 . once the processes of nitrogen fixation and nitrification have been carried out, plants are able to absorb no3 by active transport. Ppt nitrogen fixation free download as powerpoint presentation (.ppt .pptx), pdf file (.pdf), text file (.txt) or view presentation slides online.
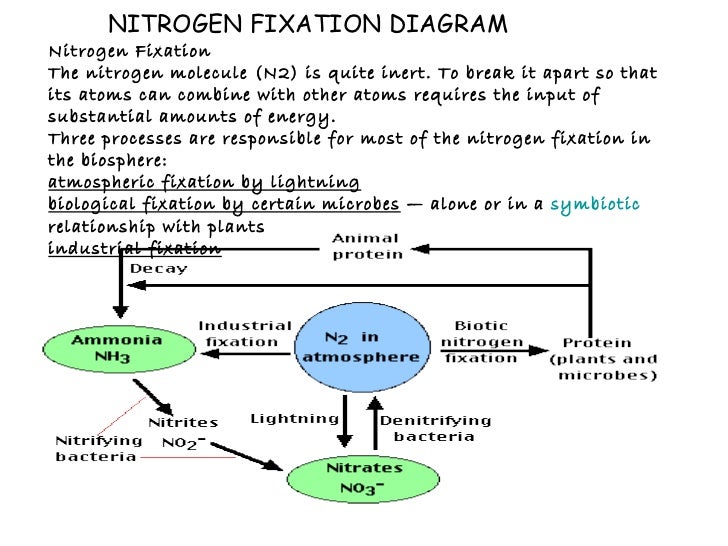
Nitrogen Fixation Cycle Assimilation plants require nitrogen in the form of dissolved nitrates no3 . once the processes of nitrogen fixation and nitrification have been carried out, plants are able to absorb no3 by active transport. Ppt nitrogen fixation free download as powerpoint presentation (.ppt .pptx), pdf file (.pdf), text file (.txt) or view presentation slides online. Learn about the essential role nitrogen plays in the biosphere, its fixation processes, and the significance of nitrogen fixing bacteria in plant symbiosis and soil fertility. Molecular nitrogen (n2) is a very common chemical compound in which two nitrogen atoms are tightly bound together. the strong triple bond between the atoms in molecular nitrogen makes this compound difficult to break apart, and thus nearly inert. Nitrogen availability often limits the primary productivity of many ecosystems, big changes in nitrogen levels lead to severe alterations of the nitrogen cycle in both aquatic and terrestrial ecosystems. Nitric and nitrous oxide can be released into the atmosphere causing potential problems. 6 nitrification it is oxidation of ammonia to nitrate (via nitrite). occurs in well drained, aerated soils by two nitrifying bacteria, nitrosomas and nitrobacter together (example of syntrophism). manure and sewage promote nitrification. nitrate is rapidly.
Comments are closed.