Moma Device Database

Moma Note: if you need help accessing information in different file formats, see instructions for downloading viewers and players. contact fda. The global unique device identification database (gudid) contains key device identification information submitted to the fda about medical devices that have unique device identifiers (udi).

Moma Device Database Devices@fda searches the following databases: premarket notifications (510 (k)s) and premarket approvals (pma). Pma data can be searched by pma number, device name, decision date, supplement type, and more. a new device that is substantially equivalent to a pma approved device means that the new device will require a pma and be classified as a class iii device. This report provides information on several frequently consulted fda medical device and drug databases. the report is intended to provide an overview of select databases to help users understand the types of information available; it is not a comprehensive listing of all current fda databases. You can use accessgudid to search for specific medical devices or download all the gudid data at once. accessgudid also offers rss feeds and apis to connect you directly to the data.

Moma Device Database This report provides information on several frequently consulted fda medical device and drug databases. the report is intended to provide an overview of select databases to help users understand the types of information available; it is not a comprehensive listing of all current fda databases. You can use accessgudid to search for specific medical devices or download all the gudid data at once. accessgudid also offers rss feeds and apis to connect you directly to the data. Enter a search term in the space below. devices@fda is a catalog of cleared and approved medical device information from fda. it includes links to the device summary information,. Fda maintains 15 medical device databases containing approval data, adverse events, manufacturer information, and regulatory guidance. the most critical are the 510 (k) database for predicate devices, pma database for class iii research, product classification for device codes, and maude for safety intelligence. Our database is synchronized with that of us fda, the most comprehensive, systematic, and updated medical device database in the us. all medical devices commercialized in the us are required to register with the fda. To modify, add, or delete information, log onto your furls account.

Moma Device Database Enter a search term in the space below. devices@fda is a catalog of cleared and approved medical device information from fda. it includes links to the device summary information,. Fda maintains 15 medical device databases containing approval data, adverse events, manufacturer information, and regulatory guidance. the most critical are the 510 (k) database for predicate devices, pma database for class iii research, product classification for device codes, and maude for safety intelligence. Our database is synchronized with that of us fda, the most comprehensive, systematic, and updated medical device database in the us. all medical devices commercialized in the us are required to register with the fda. To modify, add, or delete information, log onto your furls account.
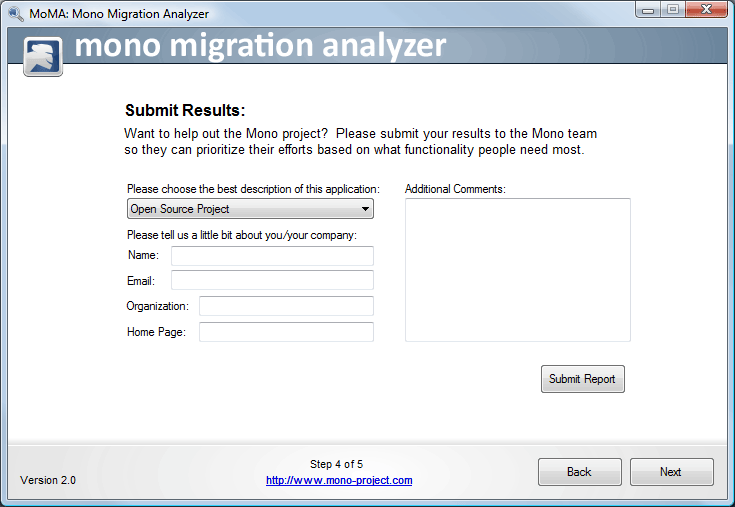
Using Moma Guide Mono Our database is synchronized with that of us fda, the most comprehensive, systematic, and updated medical device database in the us. all medical devices commercialized in the us are required to register with the fda. To modify, add, or delete information, log onto your furls account.

Order Moma
Comments are closed.