Molality Equation
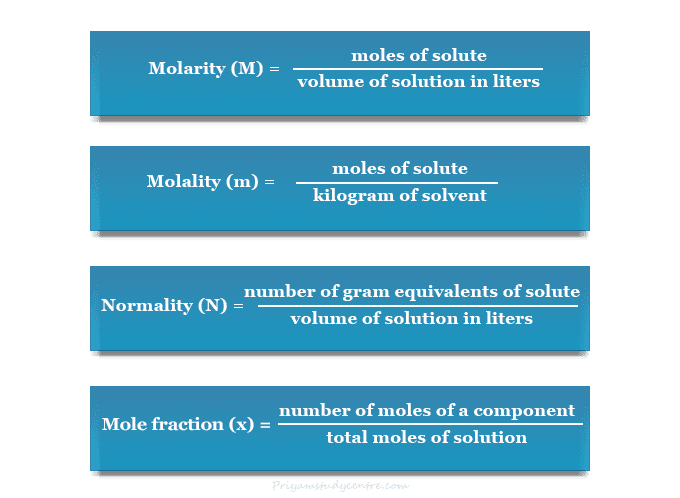
Molality Equation Learn what molality is in chemistry, how to calculate it, its advantages over molarity, and when to use it. includes examples, tips, and faqs. The molality (m) of a solution is the moles of solute divided by the kilograms of solvent. a solution that contains 1.0 mol of nacl dissolved into 1.0 kg of water is a "one molal" solution of sodium chloride.

Molality Equation Molality of a solution can be calculated by using the molality formula and the given data about the mass of solute and solvent. to calculate molality, first the number of moles of solute is determined, and then it is divided by the mass of solvent in kilograms. Molality is a measure of the concentration of a solution based on the mass of the solvent. learn how to calculate molality using the formula, its advantages and disadvantages, and compare it with molarity. In chemistry, molality is a measure of the amount of solute in a solution relative to a given mass of solvent. this contrasts with the definition of molarity which is based on a given volume of solution. a commonly used unit for molality is moles of solute per kilogram solvent (mol kg). Molality is the number of moles of solute per kilogram of solvent in a solution. learn how to calculate molality, molarity, and mole fraction using the formula and examples from general chemistry.

Molality Equation In chemistry, molality is a measure of the amount of solute in a solution relative to a given mass of solvent. this contrasts with the definition of molarity which is based on a given volume of solution. a commonly used unit for molality is moles of solute per kilogram solvent (mol kg). Molality is the number of moles of solute per kilogram of solvent in a solution. learn how to calculate molality, molarity, and mole fraction using the formula and examples from general chemistry. Learn how to calculate molality, a measure of concentration that depends only on the mass of solvent and is independent of temperature and pressure. see examples, compare with molarity, and find further readings on solutions and colligative properties. Learn what molality means in chemistry. find the formula, examples, and see how it differs from molarity. quick revision and student friendly tips included!. Learn how to calculate molality, a unit of measure for solute concentration in a solution, using the formula m = moles of solute kg of solvent. find out how molality differs from molarity and why it is useful for chemical reactions. Molality is defined as the “total moles of a solute contained in a kilogram of a solvent.” molality is also known as molal concentration. it is a measure of solute concentration in a solution. the solution is composed of two components; solute and solvent.

Molality Equation Learn how to calculate molality, a measure of concentration that depends only on the mass of solvent and is independent of temperature and pressure. see examples, compare with molarity, and find further readings on solutions and colligative properties. Learn what molality means in chemistry. find the formula, examples, and see how it differs from molarity. quick revision and student friendly tips included!. Learn how to calculate molality, a unit of measure for solute concentration in a solution, using the formula m = moles of solute kg of solvent. find out how molality differs from molarity and why it is useful for chemical reactions. Molality is defined as the “total moles of a solute contained in a kilogram of a solvent.” molality is also known as molal concentration. it is a measure of solute concentration in a solution. the solution is composed of two components; solute and solvent.
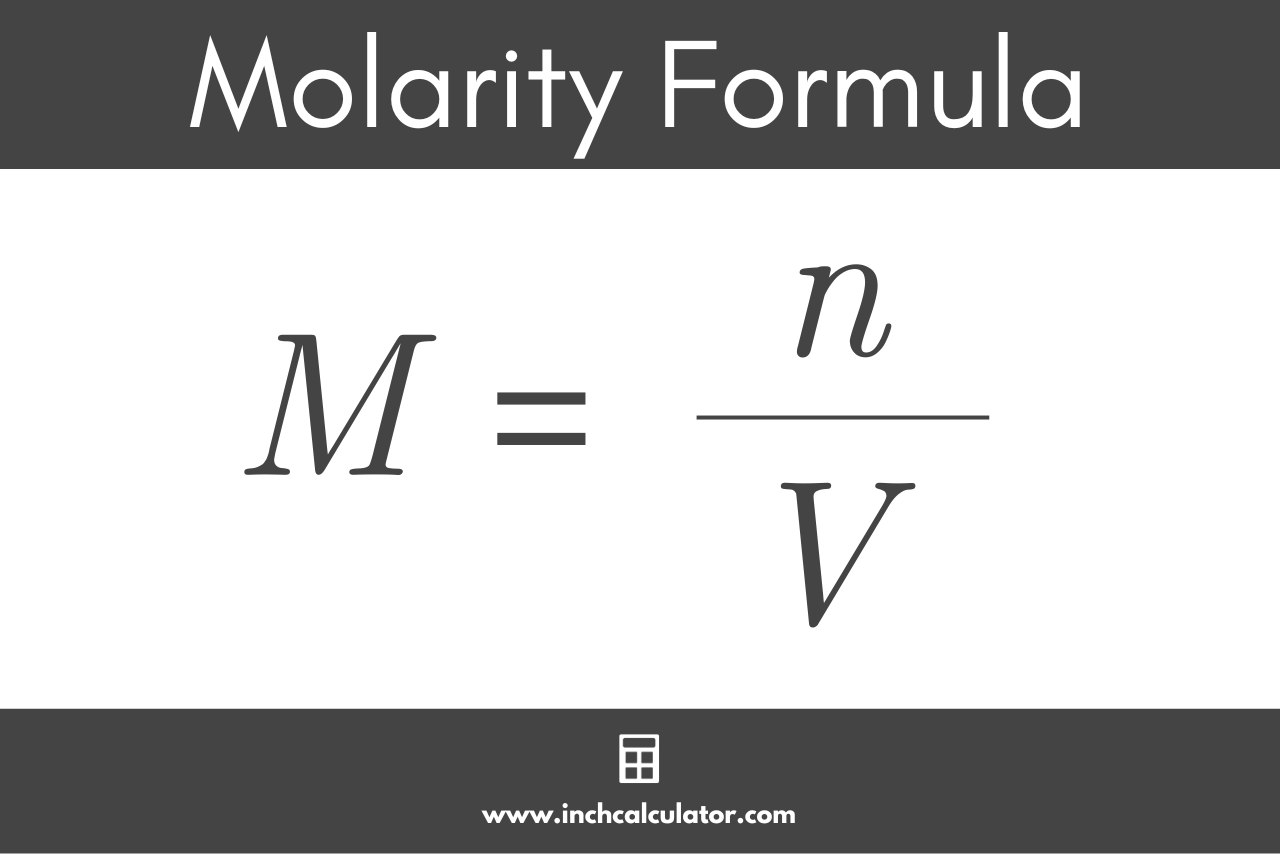
Molality Equation Learn how to calculate molality, a unit of measure for solute concentration in a solution, using the formula m = moles of solute kg of solvent. find out how molality differs from molarity and why it is useful for chemical reactions. Molality is defined as the “total moles of a solute contained in a kilogram of a solvent.” molality is also known as molal concentration. it is a measure of solute concentration in a solution. the solution is composed of two components; solute and solvent.
Comments are closed.