Mixed Solvent Recrystallization Technique

Solved When Using A Mixed Solvent For Recrystallization The Chegg In this technique, the “solvent” (i.e. the solvent which readily dissolves the compound) is used first. the compound is dissolved in the minimum volume of hot solvent. the “antisolvent” (i.e. the solvent which doesn’t dissolve the compound) is then added, carefully, dropwise. Use a glass stirring rod to scratch the flask and initiate crystallization if necessary. place the crystals in an ice water bath for 10 20 minutes and collect the solid by suction filtration.

Solved This Experiment Used A Mixed Solvent Chegg Two solvent method: the first recrystallization solvent will dissolve the compound at all temperatures. the second solvent will not dissolve the compound at any temperature. In the usual mixed solvent recrystallization procedure, dissolve your sample in a sufficient quantity of your “better” solvent by heating it up to the boiling point. There are few empirical rules that can be used as a guide to select a proper solvent, but for the most parts, the selection of the optimal recrystallization solvent is a trial and error method. Recrystallization (chemistry) recrystallization is a broad class of chemical purification techniques characterized by the dissolution of an impure sample in a solvent or solvent mixture, followed by some change in conditions that encourages the formation of pure isolate as solid crystals. [1].

Solved Mixed Solvent Recrystallization Is Useful If A Solid Chegg There are few empirical rules that can be used as a guide to select a proper solvent, but for the most parts, the selection of the optimal recrystallization solvent is a trial and error method. Recrystallization (chemistry) recrystallization is a broad class of chemical purification techniques characterized by the dissolution of an impure sample in a solvent or solvent mixture, followed by some change in conditions that encourages the formation of pure isolate as solid crystals. [1]. Solid organic compounds are usually purified by the process of recrystallization from a suitable solvent or a mixture of solvents. this process of purification is based on differential solubility of solid organic compounds and their impurities in a given solvent. The solubility of a particular solute in a solvent depends upon its polarities and the interactions with one another and temperature. the difference in solubilities of a solute in a given solvent at different temperatures is the key feature that is used for recrystallization of the solute. Transfer the material to a 50 ml erlenmeyer flask equipped with a stir bar. add about 20 ml of the solvent (determined in part i) and heat to boiling on a stir hot plate. remove any insoluble impurities by filtration, and recrystallize your product— see two solvent recrystallization guide. This paper outlines the basic principles and procedures of mixed solvent recrystallization, clarifies key steps including solvent selection, decolorization, hot filtration, and crystallization.

261 Exp 2 Mixed Solvent Recrystallization Pdf Experiment 2 Mixed Solid organic compounds are usually purified by the process of recrystallization from a suitable solvent or a mixture of solvents. this process of purification is based on differential solubility of solid organic compounds and their impurities in a given solvent. The solubility of a particular solute in a solvent depends upon its polarities and the interactions with one another and temperature. the difference in solubilities of a solute in a given solvent at different temperatures is the key feature that is used for recrystallization of the solute. Transfer the material to a 50 ml erlenmeyer flask equipped with a stir bar. add about 20 ml of the solvent (determined in part i) and heat to boiling on a stir hot plate. remove any insoluble impurities by filtration, and recrystallize your product— see two solvent recrystallization guide. This paper outlines the basic principles and procedures of mixed solvent recrystallization, clarifies key steps including solvent selection, decolorization, hot filtration, and crystallization.

Solved When Carrying Out A Mixed Solvent Recrystallization Chegg Transfer the material to a 50 ml erlenmeyer flask equipped with a stir bar. add about 20 ml of the solvent (determined in part i) and heat to boiling on a stir hot plate. remove any insoluble impurities by filtration, and recrystallize your product— see two solvent recrystallization guide. This paper outlines the basic principles and procedures of mixed solvent recrystallization, clarifies key steps including solvent selection, decolorization, hot filtration, and crystallization.
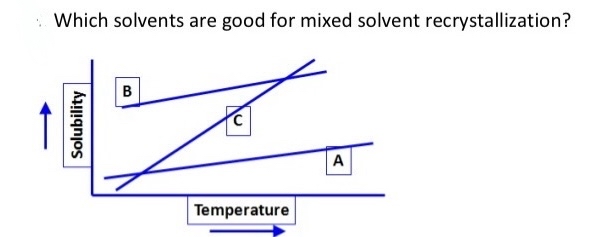
Solved Which Solvents Are Good For Mixed Solvent Chegg
Comments are closed.