Millikan Atomic Theory

Robert Millikan Atomic Theory Deeprolf Robert millikan, american physicist honored with the nobel prize for physics in 1923 for his study of the elementary electronic charge and the photoelectric effect. he is also known for his famous oil drop experiment that led to his nobel winning discoveries. When albert einstein published his 1905 paper on the particle theory of light, millikan was convinced that it had to be wrong, because of the vast body of evidence that had already shown that light was a wave.
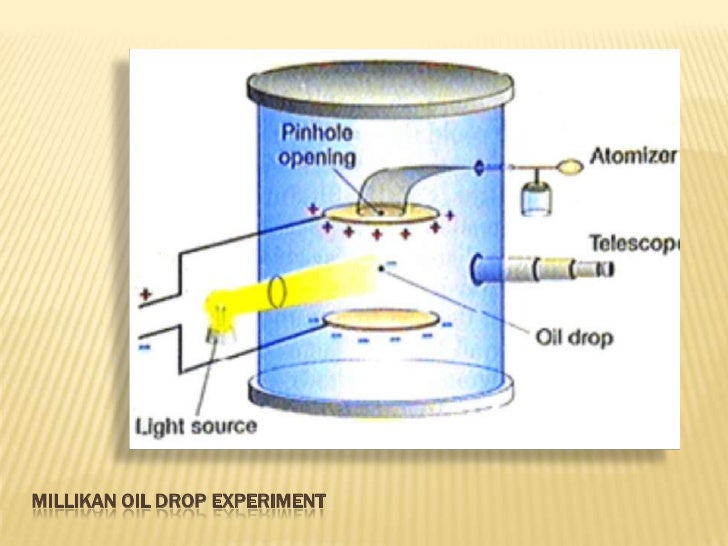
Millikan Atomic Theory Robert millikan measured the charge of the electron by studying oil drops in an electric field. he also worked on the photoelectric effect and helped build caltech into a leading physics institution. Robert millikan’s work in the early 20th century provided foundational evidence for the modern understanding of the atom. his most significant contribution to atomic theory was the determination of the precise value of the electric charge carried by a single electron. Robert millikan’s primary contribution to atomic theory came as a result of his oil drop experiment, which measured an electron’s charge. previously, j.j. thomson had suggested the hypothesis that an electron’s mass was at least 1,000 times less than the mass of the tiniest atom. The oil drop experiment was performed by the american physicist robert a millikan in 1909 to measure the electric charge carried by an electron. their original experiment, or any modifications thereof to reach the same goal, are termed as oil drop experiments, in general. what is the oil drop experiment?.

Millikan Atomic Theory Robert millikan’s primary contribution to atomic theory came as a result of his oil drop experiment, which measured an electron’s charge. previously, j.j. thomson had suggested the hypothesis that an electron’s mass was at least 1,000 times less than the mass of the tiniest atom. The oil drop experiment was performed by the american physicist robert a millikan in 1909 to measure the electric charge carried by an electron. their original experiment, or any modifications thereof to reach the same goal, are termed as oil drop experiments, in general. what is the oil drop experiment?. Scientists had now established that the atom was not indivisible as dalton had believed, and due to the work of thomson, millikan, and others, the charge and mass of the negative, subatomic particles—the electrons—were known. Learn what robert millikan discovered and the contributions of robert millikan to atomic theory. explore the oil drop experiment and its insights on the electron. Each of millikan’s contributions, while seemingly disparate, intertwined with the development of atomic theory. the table below illustrates the specific impact of each area on our understanding of atomic structure and behavior. An atomic version of the millikan oil drop experiment is performed computationally. it is shown that for planar molecules, the atomic version of the millikan experiment can be used to define an atomic partial charge that is free from charge flow contributions.

Robert Millikan Contribution To Atomic Theory Neryswap Scientists had now established that the atom was not indivisible as dalton had believed, and due to the work of thomson, millikan, and others, the charge and mass of the negative, subatomic particles—the electrons—were known. Learn what robert millikan discovered and the contributions of robert millikan to atomic theory. explore the oil drop experiment and its insights on the electron. Each of millikan’s contributions, while seemingly disparate, intertwined with the development of atomic theory. the table below illustrates the specific impact of each area on our understanding of atomic structure and behavior. An atomic version of the millikan oil drop experiment is performed computationally. it is shown that for planar molecules, the atomic version of the millikan experiment can be used to define an atomic partial charge that is free from charge flow contributions.
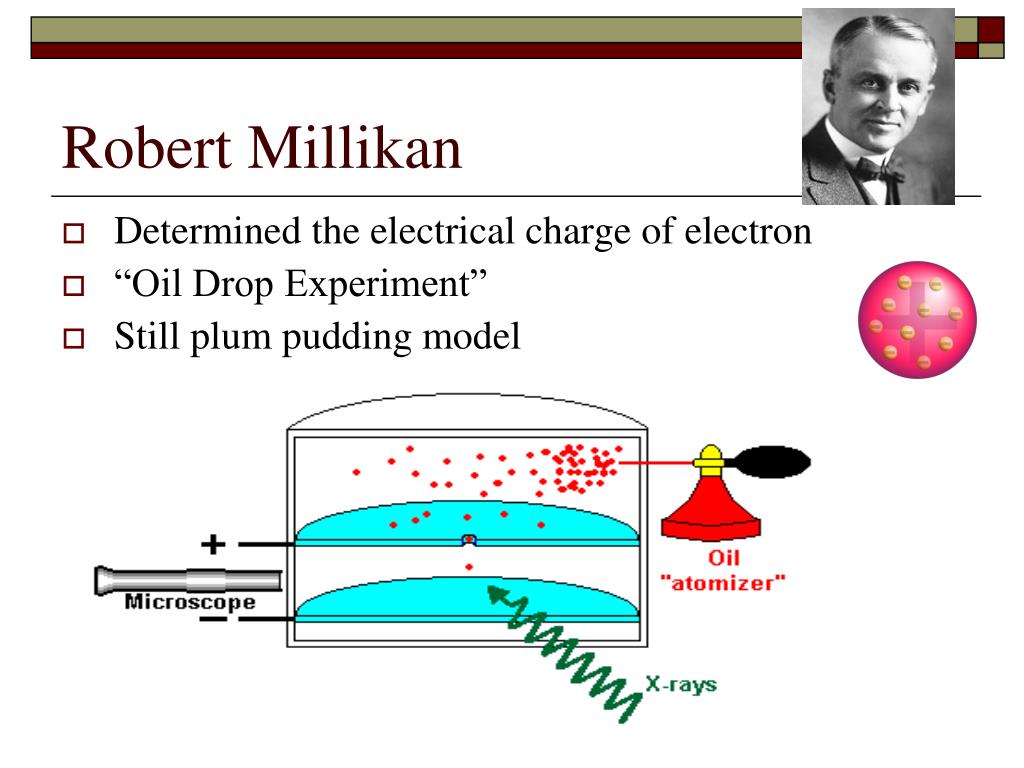
Robert Millikan Contribution To Atomic Theory Fitycq Each of millikan’s contributions, while seemingly disparate, intertwined with the development of atomic theory. the table below illustrates the specific impact of each area on our understanding of atomic structure and behavior. An atomic version of the millikan oil drop experiment is performed computationally. it is shown that for planar molecules, the atomic version of the millikan experiment can be used to define an atomic partial charge that is free from charge flow contributions.
Comments are closed.