Medical Device Development Lifecycle

Medical Device Development Lifecycle Pdf Overall, the tplc approach allows the fda to review and monitor medical devices throughout their life cycle by taking into account all available information on safety and effectiveness. Breaks down all phases of the medical design and development cycle into actionable steps. defines must know regulatory requirements for medical devices by fda, iso and iec. exposes common pitfalls and how to avoid them; detailed success strategies; and best practices for complete documentation.

Medical Device Development Lifecycle Pdf Discover the five phases of the medical device lifecycle, from concept to post market follow up, and learn how to meet regulatory and quality requirements. The medical device lifecycle covers concept, design, verification, validation, manufacturing, market launch, post market surveillance and retirement, each with specific regulatory, risk and quality management expectations. Explore the 5 phases of medical device development — from discovery and concept to fda review and post market surveillance — including key standards, regulatory requirements, and compliance guidelines. Understanding the 5 phases of medical device development and what is involved at each step. learn tips and best practices for getting a device to market.

Medical Device Development Lifecycle Pdf Explore the 5 phases of medical device development — from discovery and concept to fda review and post market surveillance — including key standards, regulatory requirements, and compliance guidelines. Understanding the 5 phases of medical device development and what is involved at each step. learn tips and best practices for getting a device to market. This guide will walk you through each phase of the medical device product development process. we'll turn what looks like a complex maze into a clear, actionable roadmap so you can move your innovation from concept to commercialization with confidence. A practical guide to the medical device development lifecycle, from concept through regulatory approval and manufacturing scale up. Objective: medical device development, from the product's conception to release to market, is very complex and relies significantly on the application of exact processes. this paper aims to provide an analysis and summary of current research in the. Medical device companies follow a well defined development lifecycle that includes concept development, design, testing, validation, and finally product launch.

Medical Device Lifecycle Overview Automation Lexoro Insights This guide will walk you through each phase of the medical device product development process. we'll turn what looks like a complex maze into a clear, actionable roadmap so you can move your innovation from concept to commercialization with confidence. A practical guide to the medical device development lifecycle, from concept through regulatory approval and manufacturing scale up. Objective: medical device development, from the product's conception to release to market, is very complex and relies significantly on the application of exact processes. this paper aims to provide an analysis and summary of current research in the. Medical device companies follow a well defined development lifecycle that includes concept development, design, testing, validation, and finally product launch.
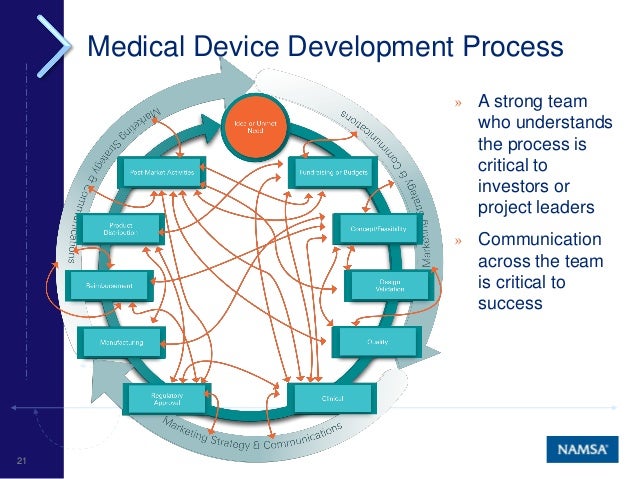
Medical Device Development Lifecycle Objective: medical device development, from the product's conception to release to market, is very complex and relies significantly on the application of exact processes. this paper aims to provide an analysis and summary of current research in the. Medical device companies follow a well defined development lifecycle that includes concept development, design, testing, validation, and finally product launch.
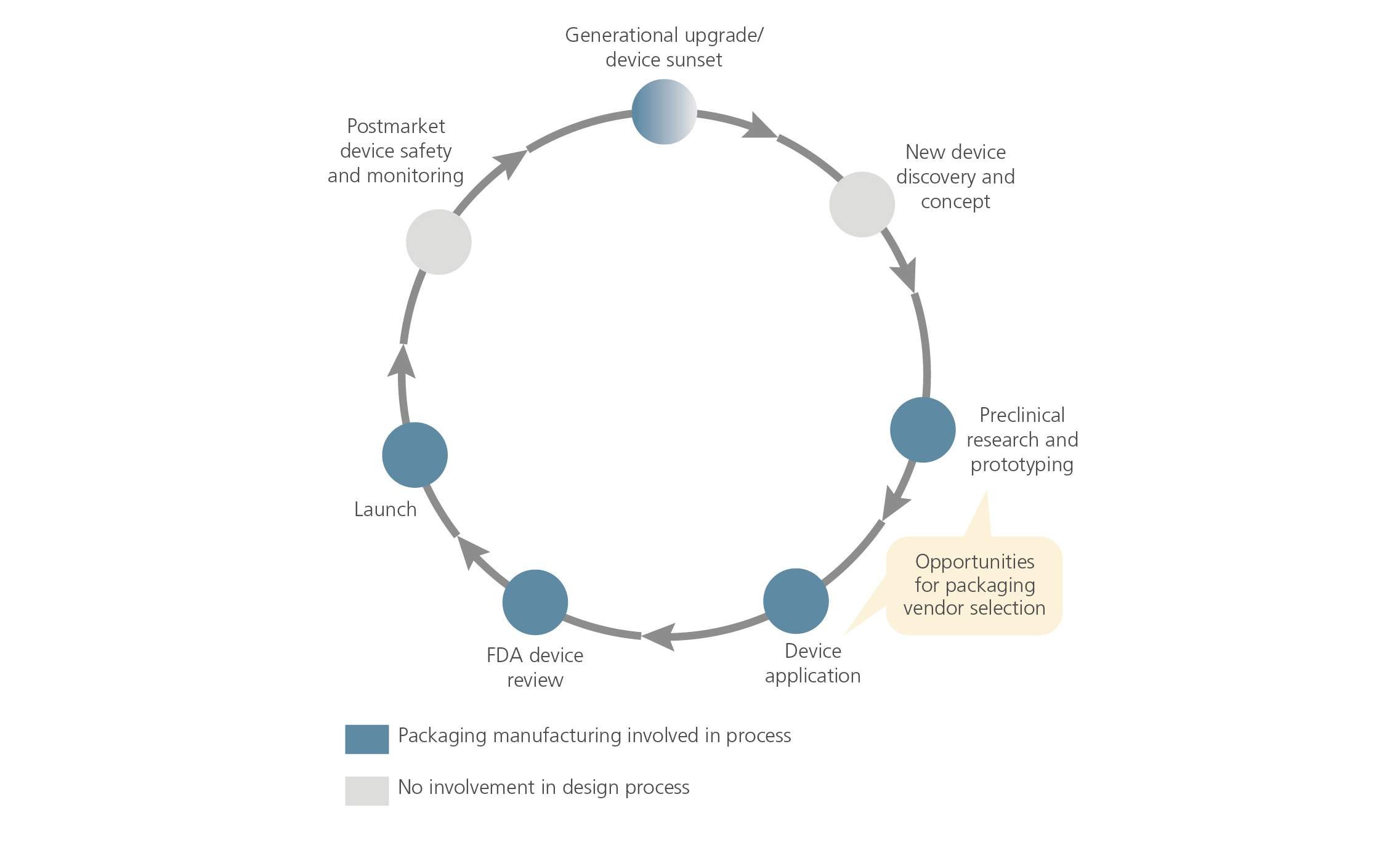
Medical Device Product Lifecycle Management
Comments are closed.